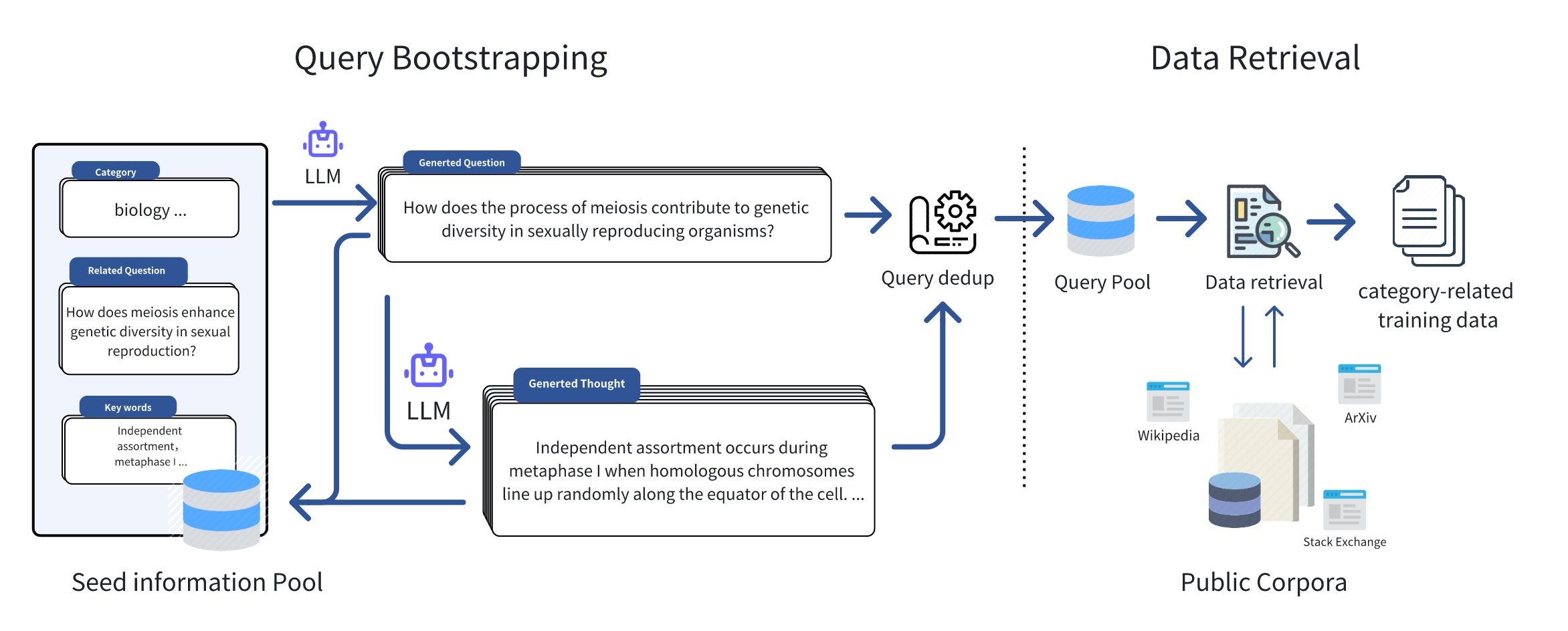
content
stringlengths 71
484k
| url
stringlengths 13
5.97k
|
|---|---|
Processing hyperspectral images allows you to decode images and recognize objects in the scene on the base of analysis of spectrums. In some problems, information about the spectra may not be sufficient. In this case, visualization of data sets may use, for object recognition, by use additional non-formalized external attributes (for example, indicating the relative position of objects). Target visualization is a visualization adapted to a specific task of application. The method discussed in this chapter uses a way to visualize a measure of similarity to the sample. As a result of the transformation, the hyperspectral (multichannel) image is converted into a single-channel synthesized image in grayscale, on which the objects of interest for the problem under consideration are selected. By changing the brightness and contrast of the synthesized image, it is possible to interactively adjust the results of automatic processing.
Hyperspectral image (HSI) is a set of images of one object (scene, slide, field of view, etc.), that match different narrow wavelength subranges, provides powerful tool for detection and recognition of objects under surveying.
Hyperspectral data use is a serious problem caused by need of processing of huge amount of information (tens and hundreds gigabytes). Most of hyperspectral image recognition algorithms are highly resource-intensive, especially when striving to provide full classification of hyperspectral information. Information about spectra may be insufficient for some tasks. In those cases acceptable interpretation cannot be realized without human assistance. The data visualization facilitates making decision for an expert-user allowing him to use unobvious signs obtained from experience. Watching all of the band images from HSI (HSI can consist of more than 300 spectral bands) is a labor-intensive and inefficient task. Segmentation methods are used to represent visual information in more compact view. These methods mark out homogeneous regions in the image that allows representing hyperspectral image in view of some synthesized image, for example, in pseudo-color, which facilitates visual perception. However, only in some cases a criterion of true segmentation exists. Visualization only allows us to reveal errors. And for the result correction, we need to process all over again using altered parameters of the algorithm. When solving a certain task, not all of the objects are of equal interest. Obviously, the visualization should be done so that the objects of interest were represented in the result image in the best way, but the details that prevent from good visual perception were eliminated. I.e. visualization should be targeted. Such adaptation of data processing to a specific practical task can be realized by means of target visualization method. As a result of such transformation the information contained in HSI is represented in the synthesized image in grayscale where the brightness range is concentrated on the details of the image that are of interest for user. The method combines mathematical computation speed with visual assessment and possibility to correct interpretation process interactively. In addition, the useful information is appreciably compressed, that is very important for remote sensing tasks.
Target visualization converts a set of images that constitute a hyperspectral image into a synthesized single-channel image that provides a visual representation of the data for a particular application. To perform the target visualization of HSI the image transform method is used. The method visualizes measure of similarity to a sample within a space of attributes [1, 2, 7].
Hyperspectral image, or so-called hypercube, is a collection of pictures of a single object that correspond to different narrow subranges of wavelengths,
where k – the spectral subrange number; (m, n) – the spatial coordinates of an image pixel; M – the number of lines in the image; N – the number of columns; Bkmn – the brightness value at point (m, n) in the kth frequency subrange.
As the attributes, that distinguish the objects of interest, the spectral characteristics are used. Moreover, can be used other characteristics that are used in tasks on image classification [3, 4], such as: features of brightness, texture, gradient and characteristics of adjacency. The values of the characteristics used for each pixel, can be obtained a priori or via image pre-processing.
Thus, each pixel of the HSI with certain spatial coordinates has its coordinates within a feature space. In the visualization method for each task on HSI processing a decision function is formed within a set of feature values. The decision function quantitatively determines belonging of the pixel to the object of interest. Then the result image is synthesized. Pixel brightness of this image conforms to the decision function value. Changing brightness range in the obtained image and using his notion about the object being studied, user can interactively set threshold to mark out the object of interest.
The main problem of the method is to choose a decision function that corresponds to a specific applied task. Some of tasks and the choice of the decision function for them are listed as an example below:
To pick out objects on the HSI that have spectral characteristics that are similar to characteristics Ok from a spectral library.
To pick out objects on the HSI that are similar to the characteristics of an object-sample with specified spatial coordinates—i.e., sample Ok is specified by spatial coordinates (m, n): Ok = Bkmn.
To pick out objects which have the most widely represented spectra on the HSI (endmembers).
To pick out objects which have the most uncommon spectra.
To pick out boundaries of homogeneous regions on the HSI.
To pick out objects on thematic images—i.e., on images obtained by using mathematical operations with spectral bands .
The suitable decision function for the tasks 1–5 is the measure of similarity to the sample [1, 2]. Similarity can be specified by one of the measures of closeness of the vectors used for HSI classification, such as:
the Euclidean distance
the distance measured by the spectral angle
the maximum distance
As a sample, multidimensional feature vector is chosen from set of points of the attribute space. A sample can be chosen either by specifying of a priori known feature values or by pointing to one of the discriminated objects.
Some set of vectors over region E of the feature space can be chosen as a sample, too. In this case it is possible to use the Mahalanobis distance—i.e., the distance given by the expression:
where —the mean of set E, and C—a covariant matrix of set E.
For task 6, the suitable decision function is
where —some function of the spectral characteristics, used when synthesizing index images, and Por—the fixed a priori value of the index.
If an information on probability distribution of attributes of the sample is available, a measure based on conditional probabilities can be used (Bayesian approach). Any of a priori estimated characteristics of studied objects can be participant in forming of a measure.
By choosing of a sample and a measure of similarity we can adapt visual representation to a specified task on interpretation. Choice of a sample can be based on:
visual analysis of some of the presented pictures;
a priori information about the object of interest location;
an information about the spectral features of the required object from a specially created database.
The value of the decision function (similarity measure value) is brightness of the pixel on the synthesized image. The largest brightness value is assigned to the pixels which are closest to the sample within the attribute space. The other pixels have brightness value according to their distance from the sample. By representing the matrix of the similarity measure in gradations of gray, we obtain a visualization of the HSI adapted to the applied task. Since the brightness of each image pixel characterizes its similarity to the object of interest, by visually choosing the brightness threshold and by varying the contrast, a specialist can discriminate the object that interests him by using his knowledge of the object and accumulated experience.
Various versions of the visualization of the same scene can be obtained by selecting various samples and similarity measures. This allows the observer to analyze the imaged scene by changing samples and similarity measures. Such a tool can be compared to a magnifying glass in feature space (on synthesized images various parts of the feature space can be detailed).
Target visualization used in various application areas of image processing, has a specificity that is determined by the area of the study and conditions of registration of images.
Computer image analysis is one of the relatively new medical technologies that are of great importance in medical research and diagnosing diseases. Due to the complexity of biomedical images, it is almost impossible to completely replace the human brain in processing them. At the same time, the method of target visualization of images allows, with the participation of a qualified user, to quickly identify and measure objects of interest.
An example of the visualization of hyperspectral images with different samples is shown in Figures 1 and 2. The source data was obtained using autofluorescent microscopy in the spectral range from 420 to 750 nm with an interval of 6 nm. (The image represents Convallaria rhizome section, acquisition with the Leica TCS SP5. The data were provided us by Biotechnologisches Zentrum der TU Dresden). Figure 1 shows four of the 50 images of the specimen as an example of the initial data.
The capabilities of visualizing multispectral images are demonstrated in Figure 2. The results obtained by transforming the 50 initial images using various samples are shown in this figure. The samples are represented in the form of graphs of spectra under the transformed images.
Another example of using visualization of hyperspectral images of biomedical theme. This example demonstrates a non-invasive method of isolation of tumor cells using a hyperspectral image. In this paper we used the HSI represented on the website of the Australian National Agency CSIRO—
In Figure 3 the multispectral image of a mouse lung with a tumor, marked with fluorescent protein, obtained in 16 spectral bands is shown. It is assumed that the tumor has uncommon spectrums relating to the attribute space. The initial multispectral image is difficult for visual analysis (it is not easy to notice differences), but on the resulting synthesized images, shown in Figure 4, various regions of the studied object are clearly distinguished. For distinguishing these regions all of the initial spectral band images are involved into processing.
Under each synthesized image in Figure 4, there is the histogram which corresponds to the distribution of distances (d) in the attribute space from the spectrum of the sample to the spectrum of each pixel in the image. The histogram provides information on the number of pixels with a spectrum similar to the spectrum of the sample. Using the histogram, it is possible to set the start threshold value for the image binarization and distinguishing the objects like the sample.
Figure 5 shows the result of selecting objects corresponding to the three samples.
The thresholds were chosen at the first minimum of the histograms.
By altering the threshold value, it is possible to vary result of the visualization in according to the contextual information and experience of the user.
The method also allows us to distinguish pixels with spectral characteristics which are rare relatively most spectra of this image. For this purpose the Mahalanobis distance for some region E can be used—i.e., the distance from the vector y to the mean vector of a set of vectors X which is specified by expression:
where is a covariant matrix of set X.
In our case, the set X is the set of vectors of the attribute space, which correspond to the most representative part of the image in Figure 6a. This area is indicated with the solid line. Figure 6b represents the result of visualization with the Mahalanobis distance over the set X use.
All pixels which are close by their spectral characteristics to have light hue. Pixels, which are appreciably different from , have dark hue. Nevertheless, the dark pixels may have different spectral characteristics. To find regions of the image, which are similar by uncommon characteristics, one should choose a dark pixel within the interesting region and use its spectral characteristics as a sample. In Figure 6c. the result of distinguishing one of the clusters of pixels with uncommon spectral characteristics is represented.
The use of contextual information when processing hyperspectral images will be shown in the example of the detection of tampering with a financial document. The HSI of the document was presented to us by ZAO “Scientific Devices”.
Figure 7 represents 4 of 125 initial pictures used for processing of the HSI of the financial document, obtained within spectral range 401–998 nm.
In these pictures, as in the others, not shown in Figure 7, it is difficult to separate the details of the text written with, though similar by spectrum, but still different ink. To detect the falsification the target visualization of the HSI consisting of 125 was used. To detect the falsification the target-visualization of the HSI consisting of 125 pictures was used. The goal of the visualization initially was to distinguish objects that have a spectrum similar to that of the main handwritten text.
The method was used; the letter o (indicated in Figure 8a by an arrow) was chosen as a specimen of the main text. The choice of this sample was based on comprehension of the task (in this part of the text the falsification was senseless). Spectral angle measure was chosen as a measure of the similarity. This gave the synthesized image shown in Figure 8b. The brightness of each pixel of the synthesized image is determined by the similarity of the spectrum that corresponds to a specified pixel with the spectrum of the sample. The image shown in Figure 8c was obtained by varying the brightness and contrast.
A section of the text was then selected that did not appear on the synthesized image (indicated in Figure 8a by an arrow). Choice of this specimen was based on the fact that the spectrum of this object was different from the main handwritten text and was handwriting, too (another sort of inks might be used). The spectrum of this section, differed from the spectrum of the main text, was taken as a sample of an forgery. Differences between spectrum of ink of the main text and one’s spectrum of the forgery are shown in Figure 9.
Visualization of the text that corresponds to this spectrum is shown in Figure 10b and after correcting the brightness and contrast—in Figure 10c. The letters “TY” and the numeral “0”belong to the forgery.
Thus, forgery detection was performed with an interactive user participation, which used information of an indirect nature (“the main text” and “handwritten text different from the main one”).
Detection of objects of interest can be performed with a greater degree of automation by combining automatic segmentation and target visualization. At the first stage, automatic segmentation of the spectra of the hyperspectral image is performed. As a result of automatic segmentation, the main spectra are determined. After such preliminary segmentation, an interactive correction can be made using the target visualization of the measure of similarity with the samples-representatives of the corrected areas.
As an example, we performed HSI processing with the joint use of automatic segmentation and the target visualization of the main spectra [6, 7, 8]. The main spectra are determined by automatic segmentation. The visualization of the similarity of the spectra of current pixels to the main spectra is shown by the synthesized images in Figure 11a. Interactive correction of these synthesized images, performed with a change in the brightness range, is shown in Figure 11b.
Efficient interpretation of images of objects and scenes may be often achieved only with combined processing of video data obtained at different time, by different surveying devices, within different spectral ranges. For combined processing of such images automatic high-accurate coordinate superposition of them is required. Such images, even being obtained with the same aspects and at the same scale, considerably differs one from other. Thus usual correlation methods of image superposition may be inadequate . There are two appreciably different approaches to image coordinate superposition: methods based on areas and methods based on image details. Each of these approaches has various advantages and disadvantages. These two approaches may be combined by means of the method of image processing by similarity to a sample .
To carry out the correlation coordinate superposition of these images, each of them is suggested to be preliminary transformed by the method of similarity to a sample involving, as an attribute space, textural, gradient features and features of adjacency.
The method does not use reference points of a scene directly. Transforming each of the superposed images by the method of similarity to a sample increase reliability of the superposition methods based on areas. Indeed, this transforming allows us to increase similarity of the images when true superposing of them and to increase their differences when relative shifting. Such transformations can be performed quite a lot, but as much as different samples exist in the fixed image.
For each sample, the correlation method determines the coordinates of the shift. The samples are chosen automatically (without the assistance of user). All of the available samples may be used. But if the images have a lot of samples, they can be selected randomly. For the selected samples a distribution is plotted.
Example of defining a coordinate shift the image obtained in the visible range with the image obtained in the infrared range, is presented in Figure 12. Presented in these shots images of the scene in the visible and IR ranges (Figure 12a and b), have parallel shift. The proposed method determined the shift; the displacement was estimated in fractions of a pixel. Figure 12c represents distribution of shifts obtained for various samples. Quality of the superposing may be estimated by statistical characteristics. For example, for this distribution of shifts the following characteristics were calculated: mode of the distribution corresponds to shifts: −0.25 pixels along OX axis, −0.5 pixels along OY axis; the mean shift along OX axis is equal to −0.2 pixels and the mean shift along OY axis is equal to −0.6 pixels; the standard deviation along OX axis is equal to 0.52 pixels and the standard deviation along OY axis is equal to 0.55 pixels.
Up-to-date equipment allows you to collect and analyze information in very narrow spectral bands and is used in various applications. In particular, for water monitoring, hyperspectral technology makes it possible to identify the distribution of algae by the concentrations of chlorophyll, and also to detect impurities of mineral substances in water and to determine the coefficients of cloudiness. Hyperspectral data can also be used to identify threatened vegetation types, some chemical toxins, changes in the chemical composition of soils, etc.
Processing hyperspectral images in remote sensing is related with the difficulties arising from the need to take into account the various distortions that appear in the process of formation of the HSI.
In known methods of processing remote sensing data before using hyperspectral information, the data pre-processing is performed. The main stages, of the pre-processing are:
radiometric correction (correcting the distortion due to the uneven sensitivity of the elements of the detectors, accounting for the effects of the atmosphere),
geometric correction (georeferencing);
combining images obtained in different spectral bands,
recalculation of data of distant shooting in values of coefficients of spectral brightness .
Such pretreatment is complex and requires additional information, time and computational resources.
Using the target visualization allows us to facilitate the processing of remote sensing data. Really, the target visualization allows us to significantly reduce influence on results of decryption of conditions of shooting (a condition of the atmosphere, exposition parameters), as a sample can be chosen on the initial images. In this case, the values of attributes of a the sample are obtained under the same conditions of shooting, as the values of attributes of the visualized scene.
Using the developed method of the target visualization the work on creation of the information layer “Swamp” on the territory of St.-Petersburg has been done . Remote sensing data, 435 micro landscapes of 11 types on total area of about 40 sq. km were mapped.
Majority of the swamps in the city is under strong anthropogenic influence due to which the images of the wetlands on the territory of St.-Petersburg are very diverse (by type) and variegated (by the spectral and spatial characteristics) structure, which creates a big problem in their interpretation by space images. As input data, the following materials were used:
the multispectral images of the territory of St. Petersburg obtained by the Quickbird satellite equipment with the resolution 2.4 m on per pixel;
the materials of field researches;
the available landscape and thematic maps.
Here, the images of the territory of town Lomonosov (near by the seaport) in four spectral bands, obtained in August by means of the Quickbird satellite equipment, are presented (Figure 13).
Using the linked archival map and the results of field researches, the necessary samples area on the image was allocated. As the sample area in this case, understand the territory which characteristics precisely match the description of this type of a micro landscape in the accepted scheme of classification.
For an example the polygon corresponding to the coastal open swamp was chosen. Due to the complexity of the research object, additional information was used in the form of additional images (Figure 14):
images derived on the base of spatial-spectral, textural, gradient and morphological characteristics of the original images;
images obtained at other season.
The result of the target visualization the coastal open swamps is presented in Figure 15a. The result of the interactive correction of the brightness and contrast are presented in Figure 15b.
The target visualization method is also applicable to the problem of finding out the boundaries of homogeneous regions on hyperspectral image.
Methods of search the boundaries are well developed for grayscale images [3, 10]. Grayscale image is considered as a function of two variables (x, y) and it is assumed that the boundaries of the regions correspond to maxima of the gradient of this function. The main problem of the search of boundary is instability their results to noise. Due to some redundancy in the joint use of multi-channel data, the selection of boundaries on the hyperspectral image is more stable to noise. The target visualization can be used to find out the boundaries of homogeneous regions. For this purpose, the hyperspectral image is transformed in the following way. For each pixel of the hyperspectral image with coordinates (m, n) the maximum spectral angle in a local region of size 3 × 3 pixels with center at (m, n) is calculated. The hyperspectral image is converted to a single-band image in accordance with the value of the spectral angle . To the obtained synthesized image in tones of gray that contains information of all channels, the known methods of search for the boundaries on halftone images can be applied. In addition, visualization of the gradient makes it possible to interactively edit the result of the boundary outlining.
Figure 16 demonstrates this method of selection of boundaries of homogeneous regions on the hyperspectral image of an urban area.
The hyperspectral image was provided by the Norwegian company Norsk Elektro Opitkk (330 channels). Figure 16a shows some shots from this hyperspectral cube. Note that the images of the individual channels are fuzzy, and some channels are highly noisy. In Figure 16b the synthesized image, visualizing the maximal spectral angles within local regions of 3 × 3, is presented. The result of the interactive selection of the boundaries on the basis of this synthesized image is shown in Figure 16c.
Use of the target visualization of hyperspectral images has a number of useful properties. Image processing by using this method is easy and quite intelligible to applied specialists. Visual data representation makes it easy for user to take a decision, allowing him to use unformalized signs according to his experience and to interactively control process of recognition of objects of interest. The transform performs rapid and compact representation of data. Indeed, measure of similarity is computed quickly and is a scalar value, that allows representing the synthesized image in one channel, and allows us to transmit it over one communication channel. It is easy enough to implement both interactive and automatic selection of objects of interest by choosing a threshold for brightness on a synthesized image. The method allows us to considerably diminish the influence of survey conditions (state of atmosphere, exposure parameters) on results of visual interpretation, because a sample can be chosen from the initial images. In this case the spectral attribute values are obtained under the same survey conditions as the feature values of the visualized scene.
The method is implemented in the basic software , which allows the target-visualization of hyperspectral images both automatically and with interactive user participation.
The program performs the following operations.
Shows the source images in each channel.
Implements a method for visualizing hyperspectral image using the method of similarity to the reference spectrum.
Changes the brightness and contrast of the visualization result.
Synthesizes a color image in a pseudocolor using different reference spectra and different similarity measures.
The program includes tools that allow you to:
Zoom the image. To do making a selection of region of an arbitrary shape on the image
Cut an arbitrary fragment of a hyperspectral image and perform the same actions with it as with the whole image.
Write the results of the processing to the file.
View the spectral curves in different, user-selected, spatial points of the image.
To store the spectra measured in the process of operation in the spectral library and use them later. | https://www.intechopen.com/books/hyperspectral-imaging-in-agriculture-food-and-environment/using-the-target-visualization-method-to-process-hyperspectral-images |
If one flower represents one season, the lotus flower, would be the one for summer. The lotus has long been seen as Hunan’s provincial flower. When taking a trip to various tourist destinations in Hunan, you will always see lotus in bloom. The flowers last from June to August, and summer is the prime time for appreciating them. Don’t miss the following places:
Loudi Tourism and Foreign Affairs Bureau and Rednet photo
Houfu Mansion, Loudi
Houfu Mansion, the former residence of Zeng Guofan (Chinese statesman, general, and Confucian scholar of the late-Qing Dynasty), is the largest and best preserved official residence in south China. Its library is among the sixth group of the cultural relics coming under state protection. The lotus in a nearly 20 hectares pond in front of the mansion bloom in June, and last for two months.
Chinese source: Hunan Tourism; Photo from Rednet
(Lotus Pond of the Village, file photo)
Sandu Town, Zixing City
Acres of lotus bloom in Sandu Town, Zixing City, Chenzhou. You may taste glutinous rice cake wrapped with lotus leaves, and also lotus tea; experience farm work like ploughing and stone milling; and, try to walk on stilts.
You may be deeply impressed by the well-preserved ancient dwellings, stone arch bridges, ancestral temples, and stilt buildings.
This year, for the first time, from July 27 to 29, a Migratory Bird Music Festival will be held. Tourists will have chances to enjoy Chinese and Western music styles and appreciate Picasso oil paintings in this vast land of lotus flowers.
Chinese source: Hunan Tourism; Photo from Rednet
Mid-July lotus pond scene by Li Maochun
Xianyu Ridge, Zhuzhou
Xianyu Ridge, about 10 km east of downtown Zhuzhou, has hundreds of mu (1 mu= 0.0667 ha.) lotus ponds, including a hundred mu of “space lotus”.
“Space lotus” is a type of lotus grown from seeds carried into space by Chinese astronauts in experiments and designed to increase their yield.
Xianyu Ridge was chosen as the main venue of the 2018 Zhuzhou Rural Summer Time Tourism Festival. There are four parts for the event: the opening ceremony; traversing the lotus pond - a TV challenge competition; Joyous lotus pond—rural music festival; energetic lotus pond—world cup carnival.
Chinese source: Hunan Tourism, Zhuzhou Tourism Website; Photo from Rednet
Photo of Junshan Wild Lotus World by Yueyang Tourism Development and Foreign Affairs Committee
Junshan Wild Lotus World, Yueyang
Walking through Junshan Wild Lotus World on Tuanhu Lake, you will find open water teeming with lotuses. It boasts the largest area of wild lotus in Asia, stretching to over 5,000 mu (about 333 hectares). Giving it its name --" Home to Chinese Wild Lotus" by the China Wild Plant Conservation Association in 2009. Boating and harvesting lotus seeds on the lake is a must-do for visitors.
Junshan Wild Lotus World is located at Guangxingzhou Town, Junshan District, Yueyang City
Chinese source: Hunan Tourism; Photo from Rednet
Hunan Provincial Botanical Garden flowers taken by Peng Wei
Hunan Provincial Botanical Garden
It boasts the most varied species of lotus flowers in Hunan, reaching almost 600. The best viewing place is close to the Science and Research center. There are many kinds of flowers such as roses, lotus, peonies, daisies and tulips. There are many types of blooms, single-petal, and multiple-petal. There are many colors red, yellow, white, reddish-yellow, etc. There are three petal flower and four petal flowers. etc.
Chinese and photo source: Rednet
Photo from icswb.com
Sujiatuo Lotus Base, Kaifu District, Changsha
Sujiatuo is the lotus garden nearest to downtown Changsha. On low-lying land with rich water resources it is suitable for lotus growing. Most growers came from Huashi Town, Xiangtan County. When summer arrives, thousands of mu of lotus flowers bloom under Sujiatuo Viaduct on Changsha’s North Second Ring Road.
Chinese source: Hunan Tourism;
Kaihui Town, Changsha County
The First Lotus Culture Tourism Festival will be launched between July 14 and September 15 in Kaihui Town, Changsha County.
Chinese and photo source: Changsha Tourism
The tiny-sized lotus flower
Water lilies
Lotus Park at Panlong Grand Amusement Park
Covering a total area of 300 mu, the lotus park inside of Xiangtan’s Panlong Grand Amusement Park is the Only Lotus Species Treasure of China. Over 30 species have won gold prizes among lotus exhibitions.
Chinese and photo source: Xiangtan Tourism Overseas Affairs
Wanhe Garden, Pushui Town, Xiangxi
Lotus flowers in Wanhe Garden are in blossom every June and July. You may also visit the local dwellings and enjoy Chenghe Gaoqiang (an upper-octave opera) performance. The millennium town of Pushi has been promoted as a national 4A-level scenic spot.
Chinese source: Hunan Tourism; Photo from: Luxi Pushi Ancient Town Culture Tourism Official Website
Photo of the blooming lotus flowers by CPC Nanxian County Committee Publicity Department
Luowen Village, Nanxian County, Yiyang City
When strolling in Luowen Village, some gorgeous walls come into sight first. The village is the first international graffiti art village in Hunan. A pavilion in the pond center provides visitors a good place to enjoy blooming lotus flowers. | http://enghunan.gov.cn/hneng/newscollection/news2018/July2018/201807/t20180727_5062809.html |
A classic Chinese writing reads "I love the lotus because while growing from mud, it is unstained." Ever since, the lotus has become a metaphor for honesty and a noble character, symbolic of maintaining ones purity even when immersed in an atmosphere of filth and corruption.
Embroidered alongside this elegant piece is the following poem:
The lotuses are like sisters, leaning on each other,
Leisurely standing beside the bridge
Making an appointment to blossom together
Later than any other flower.
But their late blossom does not affect their rank
As the most beautiful flowers in the garden.
- Frame: 29" x 21", Mat Opening: 23" x 15"
- Premium solid wood frame, black matte finish
- High-clarity glass
- Bevel-cut raised white mat
- Soft-mount option
Framed Embroidery: To clean, wipe with a soft cloth. To hang, use the preinstalled hangers on the back of the frame.
Soft-Mount Embroidery: Enjoy as-is or frame to your liking. The soft-mount can be cut to size and attached to a backboard. | https://www.artofsilk.com/collections/framed-artwork/products/lotus-flowers |
The lotus is one of the most significant symbolic flowers in the Egyptian-Nubian culture. It symbolizes birth and life after death. Our jewelry is inspired by a lotus pendant with a center lotus flower made by gold wire and glass flux. It is the only piece of its kind to be found together with numerous other pieces of jewelry in the tomb of the Meroitic queen Amanishakheto, who is believed to have ruled from 10 BC to 1 AD.
Materials: bronze with gold finish and epoxy, snake chain included
Pendant measures approximately 1" x 1.25"
Chain Length: 18" | https://shop.spacedustla.com/products/egyptian-lotus-necklace |
Between the golden-yellow lotus leaves and stalks, we see swaying white lotus flowers with thick black border lines and red fish swimming among them. The transcendent serenity depicted highlights the infinite vitality of life.
- 12 inches
- Bone china
Tai-Hwa is a Taiwanese ceramics brand whose mission is to combine beauty and usefulness. Their distinctive pottery fuses traditional techniques with cutting-edge experimentation, resulting in vivid colors and unrivaled quality that will last a lifetime. After 30 years in business, Tai-Hwa still produces in the old pottery district of Yingge, in Taipei, where local artists use local materials to bring Taiwanese pottery to the world. | https://azneo.com/products/lotus-12-bone-china-gold-transfer-round-plate |
This chapter serves as a fantastic test preparation tool for individuals who want to better understand or get reacquainted with chemical bonds and reactions. Enhance your comprehension of the laws of thermodynamics, redox reactions, different types of chemical bonds and more. In no time, you will be able to:
- Define covalent and ionic chemical bonds
- Describe polar covalent and hydrogen chemical bonds
- List and discuss basic properties of chemical reactions
- Provide examples of hydrolysis and dehydration
- Share the definition of ionic compounds
- Differentiate between anabolism and catabolism
- Discuss weak acids, weak bases and buffers
Access the lessons in this chapter as short videos that average about 8 minutes each, or scroll down to view full transcripts you can read online or print out. Each lesson is accompanied by a multiple-choice quiz designed to test your knowledge of the concepts it covers. Clarify any topics you don't fully understand by sending your questions to our experts. When ready, gauge your overall knowledge of chemical bonds and reactions using our practice chapter exam.
1. Chemical Bonds I: Covalent
Mom always said that sharing is caring. This lesson will explore how electrons affect the chemical reactivity of atoms and specifically the merits of sharing electrons.
2. Chemical Bonds II: Ionic
Did you know that the scientific name for table salt is sodium chloride? Find out how sodium and chlorine atoms come together to form your favorite seasoning.
3. Chemical Bonds III: Polar Covalent
Are you confused about how you can tell what kind of bond two atoms will form? This lesson will help you understand the difference between polar and nonpolar covalent bonds as well as how to predict how two atoms will interact.
4. Chemical Bonds IV: Hydrogen
This lesson defines and discusses important concepts behind hydrogen bonding. You'll learn when and why these bonds occur and which atoms are often involved.
5. The Laws of Thermodynamics
Learn about the first and second laws of thermodynamics. Find out how energy is generated, how it converts from one form to another, and what happens to energy in a closed system.
6. Basic Properties of Chemical Reactions
Learn how about the various components of a chemical reaction, and how those components function. Use this lesson to understand the basic properties of different kinds of chemical reactions.
7. Redox (Oxidation-Reduction) Reactions: Definitions and Examples
This short video will explain oxidation-reduction reactions, or redox reactions for short. The focus is on how electrons are transferred during redox reactions. Learn some neat mnemonic devices to help you remember when an atom is oxidizing or reducing.
8. Hydrolysis and Dehydration: Definitions & Examples
Water is an important component of cellular processes. Two of these processes, dehydration and hydrolysis, help your body build large molecules from small ones and break down large ones into usable components.
9. What Are Ionic Compounds? - Definition, Examples & Reactions
Ionic compounds are a common, yet special type of chemical compound. In this video lesson, you will learn about their formation and structure and see examples of compounds formed by ions.
10. Anabolism and Catabolism: Definitions & Examples
Metabolism breaks down large molecules like food into usable energy. This energy drives bodily processes critical to survival. In this video lesson, you will learn about the two forms of metabolism that break down and build up molecules and see examples of each.
11. Weak Acids, Weak Bases, and Buffers
This lesson covers both strong and weak acids and bases, using human blood as an example for the discussion. Other concepts discussed included conjugate acids and bases, the acidity constant, and buffer systems within the blood.
Earning College Credit
Did you know… We have over 200 college courses that prepare you to earn credit by exam that is accepted by over 1,500 colleges and universities. You can test out of the first two years of college and save thousands off your degree. Anyone can earn credit-by-exam regardless of age or education level.
To learn more, visit our Earning Credit Page
Transferring credit to the school of your choice
Not sure what college you want to attend yet? Study.com has thousands of articles about every imaginable degree, area of study and career path that can help you find the school that's right for you. | https://study.com/academy/topic/chemical-bonds-reactions.html |
NOBCChE Collaborative is proud to announce our Faculty Seminar Series, highlighting faculty’s scientific expertise and covering a wide range of topics. Each seminar is free and open to the public, but registration is required. Use the button above to register.
October 28th - Psaras McGrier, PhD
Title: Synthesis and Design of Functional Covalent Organic Frameworks
Abstract: Covalent organic frameworks (COFs) are an advanced class of crystalline porous polymers that are composed of light elements (C, H, O, N, and B) linked via strong covalent bonds. COFs are generally constructed by using reversible bond forming reactions to yield crystalline materials with high surface areas, low densities, and high thermal stabilities. These features make COFs useful for carbon capture, gas storage, and sensory applications. This lecture will discuss employing a bottom-up approach to create novel functional COFs that can bind small metal cations (e.g., Li, Ca, etc.), and some low-oxidation state transition metals (e.g., Ni(0), Co(II), Fe(II), etc.). The prospect of utilizing these COFs for applications related to gas separations, catalysis, and energy storage will be highlighted.
November 18th - Peter Chen, PhD
Title: TBD
Abstract: TBD
December 16th - Gabriela Schlau-Cohen, PhD
Title: Why don't plants get sunburn?
Abstract: In green plants, chlorophyll-containing proteins known as light-harvesting complexes (LHCs) capture solar energy and feed it to downstream molecular machinery. Under high light (i.e., sunny days), excess absorbed energy can cause damage. Thus, LHCs have evolved a feedback loop that triggers photoprotective energy dissipation, solving the so-called “intermittency problem” in solar energy. A long-standing proposal has been that conformational changes of the LHCs activate dissipative photophysical pathways among the chlorophyll. First, we use single-molecule spectroscopy to identify the conformational states of the LHCs, uncovering parallel conformational dynamics that regulate fast and slow changes in sunlight. Second, we use 2D electronic spectroscopy to map out the corresponding photophysics, revealing dissipative chlorophyll-to-carotenoid energy transfer, a hypothesized yet previously unobserved pathway. Collectively, these multi-timescale measurements elucidate the multi-timescale dynamics of photoprotection.
January 27th - Mary Jo Ondrechen, PhD
Title: How do enzymes impart catalytic superpowers to amino acids?
Abstract: Enzymes catalyze reactions under mild conditions that would otherwise require extreme conditions such as high temperature or strong acidity or basicity. To achieve this, enzymes often impart reactive chemical properties to amino acid sidechains that are far less reactive in the absence of the protein environment. In the enzymatic environment, active site amino acid sidechains that are weak Brønsted acids or bases in a small peptide can transform into a strong acid or base. The primary amine side chain of lysine, which would normally be protonated at neutral pH, can be deprotonated to serve as a nucleophile. We present a set of 20 enzymes that represent all six major EC classes and a variety of fold types for which experimental studies of the catalytic residues' mechanistic roles have been reported in the literature. For these 20 enzymes the computed electrostatic and proton transfer properties are investigated. The catalytic aspartates and glutamates are shown to be strongly coupled to at least one other aspartate or glutamate residue, and frequently to multiple other carboxylate residues, with intrinsic pKa differences less than ~1 pH unit. These catalytic acidic residues are sometimes coupled to a histidine, wherein the intrinsic pKa of the acid is higher than that of the His. Anion-forming residues, Tyr or Cys, with intrinsic pKa higher than that of the lysine, are found strongly coupled to all catalytic lysines in the set. Some catalytic lysines are also coupled to other lysines with intrinsic pKas within ~1 pH unit. Some basic principles about the design of enzyme active sites are discussed. The interactions described here provide important clues about how side chain functional groups that are weak Brønsted acids or bases for the free amino acid can become strong acids, bases, or nucleophiles in the enzymatic environment. Supported by NSF CHE-1905214.
February 24th - Stephen Leffler Buchwald, PhD
Title: Palladium-Catalyzed Carbon-Heteroatom Bond Formation
Description: The history of the development of Pd-Catalyzed Carbon-Heteroatom Bond Formation as well as a description of the basic mechanistic considerations, recent examples and an application to the modification of proteins will be described.
March 31st - Christine Thomas, PhD
Title: Incorporating Metal-Ligand and Metal-Metal Cooperativity into Catalysis
Abstract: The formation and cleavage of chemical bonds in catalytic reactions relies on accessible two-electron redox processes that are often challenging for base metals such as first row and early transition metals. Metal-ligand and metal-metal cooperativity provide a potential solution to this challenge by enabling heterolytic bond cleavage processes and/or facilitating redox processes. Both strategies will be discussed, showcasing the many ways that metal-ligand and bimetallic cooperativity can operate and the methods by which cooperativity can be built into catalyst design. A tridentate pincer ligand featuring a reactive N-heterocyclic phosphido fragment is found to be both redox active and an active participant in bond activation across the metal-phosphide bond, with catalytic applications in alkene hydroboration. A tetradentate bis(amido)bis(phosphide) ligand has been coordinated to iron and it has been shown that the resulting complex can activate two σ bonds across the two iron-amide bonds in the molecule without requiring a change in the formal metal oxidation state. In the context of metal-metal cooperativity, phosphinoamide-linked early/late heterobimetallic frameworks have been shown to support metal-metal multiple bonds and facilitate redox processes across a broad range of metal-metal combinations and the resulting complexes have been shown to activate small molecules and catalyze organic transformations. | https://www.nobcche.org/collaborative-faculty-seminar-series |
Enzymes are protein catalysts that are responsible for lowering the energy barrier of many biological reactions. They function by reducing the activation energy of the reactions that they catalyse which allows the metabolic reaction to occur faster and at physiological temperatures. Enzymes are specific for one substrate due to the unique arrangement of their active sites. However many enzymes cannot function on their own, and instead require non-protein cofactors. Enzymes that require such cofactors are called apoenzymes. When the cofactor and the apoenzyme are combined a holoenzyme is produced, this holoenzyme is able to catalyse reactions.
Cofactors function by providing functional groups needed for the reaction or by slightly altering the structural conformation of the enzyme to which they are bound. This alteration allows substrates to bind more easily, making a reaction more probable. Cofactors can be prosthetic groups which are tightly bound to the enzyme, or the cofactors can be coenzymes which can be loosely bound. Coenzymes are usually released from the active site of the enzyme following a reaction.
Coenzymes are a type of cofactor and they are bound to enzyme active sites to aid with their proper functioning. Coenzymes which are directly involved and altered in the course of chemical reactions are considered to be a type of secondary substrate. This is because they are chemically changed as a result of the reaction unlike enzymes. However unlike the primary substrates, coenzymes can be used by a number of different enzymes and as such are not specific. For example hundreds of enzymes are able to use the coenzyme NAD.
The function of coenzymes is to transport groups between enzymes. Chemical groups include hydride ions which are carried by coenzymes such as NAD, phosphate groups which are carried by coenzymes such as ATP and acetyl groups which are carried by coenzymes such as coenzyme A. Coenzymes which lose or gain these chemical groups in the course of the reaction are often reformed in the same metabolic pathway. For example NAD+ used in glycolysis and the citric acid cycle is replaced in the electron transport chain of respiration.
Due to the importance of coenzymes in chemical reactions, and due to the fact that they are used up and chemically altered by reactions, coenzymes must be continually regenerated. For example, synthesis of B vitamins is a complex, step wise process because the B vitamins have chiral centres which are complicated to synthesise. Coenzymes that are produced from B vitamins are especially important to the proper functioning of enzymes involved with regulation of metabolism and with the release of energy from food. Important B vitamins that are used as large components of coenzymes include riboflavin, niacin, biotin, pantothenic acid, B6, folate and B12. For example riboflavin, or vitamin B2 is used as a large component of FAD and FADH, and niacin is an important component of NAD and NADH.
However vitamins cannot be made by the body, but instead they must be consumed in the diet. Therefore vitamins are essential components of the diet. Although the human body uses more than its own body weight in ATP, not as much of the vitamin that is used to produce the coenzyme is needed to be consumed. This is because the body is able to use the vitamins very instensively through regeneration.
The function of ATP is to transport chemical energy within cells for metabolism, and as such ATP is often referred to as the energy currency of cells.
Adenosine triphosphate is composed of an adenine nucleotide base, a ribose sugar and three phosphate groups. Energy can be released from ATP when the terminal phosphate group is released in a hydrolysis reaction. This is because the energy of ATP is held in the bonds between the phosphate groups and when the bonds are broken it is accompanied by a release of energy.
NAD is composed of two nucleotides, adenine and nicotinamide. The nucleotides are held together by a pair of phosphate groups which act as a bridge and are also bonded to a ribose sugar each. The function of NAD is to carry electrons from one enzyme controlled reaction to another. As such NAD is involved with redox reactions because substrates are either oxidised, in which they lose electrons or are reduced in which they gain electrons. NAD is either found as NAD+, which is an oxidising agent and is involved with accepting electrons from other molecules, or NADH which is used as a reducing agent to donate electrons to other molecules.
FAD is composed of an adenine nucleotide, a ribose sugar and two phosphate groups. FAD can also exist as a monophosphate and is called flavin adenine monophosphate (FMN). The primary role of FAD is in oxidative phosphorylation. FAD is involved with redox reactions and like NAD, FAD can exist in two redox states; FAD and FADH. The two states are interconvertable as a result of the addition or removal of electrons.
This is possible because FAD is able to accept hydride ions with their electron pairs. For example, FAD is a coenzyme used by the enzyme succinate dehydrogenase to help catalyse a reaction. The role of FAD in this reaction is to accept two electrons from succinate which results in the production of fumarate. FAD is reduced to FADH2 but remains tightly bound to succinate dehydrogenase. No further reactions can occur until FAD is regenerated.
Coenzyme A is not tightly bound to the enzymes to which it is associated and is able to freely be released. It plays an important role in the metabolism of protiens, carbohydrates and fats which are important reactions that allow the energy from food to be released. For example coenzyme A is required for the oxidation of pyruvate in the citric acid cycle.
In addition coenzyme A is involved with acetylation reactions. These reactions are important in proper protein function and as a result many of the proteins in the body have undergone such modification reactions in which an acetate group is added to the protein. The acetate group is donated from coenzyme A. The addition of an acetate group alters the 3D structure of the protiens to which it is added and as a result thier function is also altered. In some cases a long chain fatty acid is also donated to the protien. This addition is needed for the cell signalling properties of various membrane protiens.
Acetate groups from coenzyme A are also added to various substrates in reactions involved with gene expression and cell division. Coenzyme A is also important in the synthesis of cholesterol and steroid hormones, and is required for the detoxification of a range of harmful drugs that can accumulate in the liver.
Alcohol dehydrogenase (ADH) is an enzyme which uses NAD+ as a coenzyme. ADH has two binding regions, one where the primary substrate, ethanol binds and one where the coenzyme, NAD+ is able to bind.
The enzyme is responsible for the conversion of ethanol to ethanal. The reaction is an oxidation- reduction reaction and results in the removal of two hydrogen ions and two electrons from ethanol. The hydrogen ions and electrons are added to NAD+ which converts the coenzyme to NADH + H+. This is the first reaction involved with the metabolism of ethanol.
Stamp, S. (2012) Alcohol dehydrogenase. Fastbleep.com Biochemistry Chapter.
Becker, W. Kleinsmith, L.J. Hardin, J. Bertoni, G.P. (2008) The World of the Cell, Benjamin Cummings.
Price N.C., Stevens L., 1999. Fundamentals of Enzymology, 3rd Ed., Oxford University Press, New York. | https://www.fastbleep.com/wiki/article/1183 |
Redox potential, a measure of how oxidising or reducing an environment is, is tightly regulated by cells to minimise detrimental chemical oxidation and reduction reactions. In proteins, it is the sulfur containing cysteine residues that can be post-translationally modified through specific redox reactions, for example, the formation of disulfide bonds between cysteine residues can be crucial to protein structure. It has recently been hypothesised that signalling pathways utilising redox regulated proteins may be arranged into electrochemical series. The characterisation of the redox properties of specific cysteine residues in proteins has proven difficult using traditional redox characterisation methods such as cyclic voltammetry. A number of biochemical methods have been developed for studying the effect of the redox environment on proteins, many making use of mass spectrometry and allowing for localisation of the site of the modification to specific cysteine residues. However, fewer methods have been reported that facilitate accurate quantification for the determination of the mid-point potential of these redox regulated cysteine residues. Here, a differential labelling protocol using high resolution mass spectrometry techniques for the study of redox chemistry of cysteine residues in proteins will be reported. The protocol exploits the novel chemistry of thiol groups for specific alkylation and allows for both qualitative and quantitative experiments. Thioredoxin-1 from E. coli and human systems was used as a model protein and a novel disulfide bond was characterised. The reducing potential of the active site cysteine residues of human thioredoxin were found to be very similar to those of the E. coli proteoform, -276 ± 1 and -281.4 ± 0.3 mV respectively. The remaining three cysteine residues of human thioredoxin were found to be regulated at more oxidising potentials. The protocol developed was applied to a protein from the cell death pathway of apoptosis; human caspase-3 is an executioner protease from the caspase cascade. Caspase-3 was found to contain three redox sensitive cysteine residues. The catalytically active cysteine residue was redox regulated via two mechanisms, glutathionylation and disulfide bond formation. One of these mechanisms gives the active site cysteine residue a calculated reducing potential of -165 ± 6 mV supporting the correlation between caspase-3 activity and its observed role in the apoptotic pathway but not in necrotic cell death. | https://era.ed.ac.uk/handle/1842/18020 |
This blog post will answer the question, “what is the difference between flammable and combustible” and cover topics like factors differentiating between flammable and combustible, and frequently asked questions related to the topic.
What is the difference between flammable and combustible?
Here are the differences between flammable and combustible:
What are Combustible Materials?
Combustible materials are those that can burn in the air with some effort and an igniting source. The flashpoints of flammable liquids vary from 60 to 93 degrees Celsius. It implies they’re less volatile and, as a result, won’t burn quickly at room temp (25-35 °C).
Combustible Materials Examples
Combustible liquids include cyclohexane formic acid, anthracene, dichlorobenzene, fats, and hydrazine.
Combustible liquids’ flashpoints and fire points are lower than their boiling points. Paper and wood are examples of flammable materials.
What are Flammable Materials?
Flammable objects are flammable substances that may quickly burn in the air if an ignition source is present. When compared to combustible materials, this indicates they have a lower flashpoint temp.
The combustible liquid’s flashpoint temperature ranges from 0 to 60 degrees Celsius. They have higher volatility than flammable materials. They’re also referred to as flammable substances.
Flammable liquids may quickly catch fire at working/room temp, hence they must be handled with caution when working.
Flammable Materials Examples
Pentane, hexane, petrol (gasoline), ethyl acetate, acetone, methanol, and isopropyl alcohol are all flammable liquids.
Metallic sodium, greasy materials, matches, and nitrocellulose substances are examples of flammable solids.
Factors that influence a material’s flammability or combustibility
The extent of flammability is determined by the material’s volatility, which is determined by the vapor pressure.
The temperature has an impact on vapor pressure. The extent of flammability is also determined by the material’s surface area.
Let’s take a closer look at the impact of these interconnected elements on the degree of flammability.
1. Volatility: The more volatile a chemical is, the greater its flammability, i.e., the more volatile compound will burn more readily than a less volatile one.
2 Vapour Pressure: Vapour pressure is the pressure imposed on the surface of a liquid by its vapor at equilibrium when the rate of vapor condensation equals the rate of liquid evaporation.
For instance, a chemical with a high vapor pressure will be more volatile and so quickly combust in the air.
3 Temperature: As the temperature increases, the kinetic energy of the molecules of a liquid rises, increasing the number of liquid molecules that enter the vapor phase.
4 Surface area: Increasing the surface area of a substance, i.e. using finely split material, might improve its flammability.
The material’s increased surface area will increase the number of vapors it produces, allowing it to burn more readily in the air.
The Hazards of Combustible and Flammable Materials
We know that combustible materials have a higher flashpoint temperature than flammable ones. However, this does not imply that combustibles are less harmful than flammables.
At temperatures above their respective flashpoints, both flammable and combustible products are utilized, transported, and stored. To avoid ignition, these materials must be handled with extreme care.
Otherwise, combustible substance vapors might readily catch fire at working temperature if the space is not properly ventilated.
When the ambient temperature rises over the flashpoints of combustible materials, they emit flammable vapors and catch fire.
In summary, flammable and combustible materials may endanger people’s lives as well as damage transportation and storage materials. It also contributes to environmental contamination.
As a result, extraordinary attention must be used while storing, transporting, and handling these substances.
What measures should be taken while keeping these materials?
Let’s have a look at these safeguards.
Precautions to take while storing flammable and combustible materials
When storing flammable and combustible substances, we, as ordinary people, must keep a few things in mind. These are the main points.
- The room where these substances are housed should have enough ventilation.
- These items should be kept away from any source of ignition.
- Flammable and combustible compounds should be kept apart from incompatible substances to avoid reactions.
When flammable and combustible liquids are mixed with incompatible substances, harmful chemical reactions such as combustion and oxidation may occur.
As a result, harmful compounds that are incompatible must be separated.
The hazardous products separation chart determines a specific separation distance between incompatible compounds for this purpose.
Precautions using combustible and flammable substances
Ensure that all storage containers are in excellent working order, that they are closed, and that they are correctly labeled.
To avoid ignitable vapor/air combinations or inhalation of harmful vapors or gases, use flammable or combustible liquids under a fume hood.
Ignition sources (hot substances, flames, or sparking devices) should not be placed near these liquids. If at all feasible, use electrical heating to substitute open flames.
Static sparks are likely to be produced by ground equipment.
When flammable or combustible substances are warmed to or above their flash points, take extra care.
Compressed or molten gases are very flammable. Refer to the EH&S Compressed Gas Guidelines for further information.
A major fire hazard exists when incompatible substances come into contact. It is necessary to follow proper storage and handling methods. Some safety methods are listed below:
- Cabinets for storing things
- Cans of Safety
- Storage in the Refrigerator
I will now elaborat eon the guidance given above.
Cabinets for storing things
At any one moment, no more than ten gallons (37.9L) of flammable materials may be stored outside of a certified storage cabinet. Flammable and flammable liquid storage cabinets must fulfill NFPA (National Fire Protection Association) requirements and may not be altered in any manner. Ventilation of storage cabinets is not advised. Storage cabinet vent apertures must be sealed with the bungs provided with the cabinet if they are not vented.
Cans of Safety
Safety cans can hold up to 5.3 gallons (20L) of flammable or combustible substances. Safety cans must be made of metal and have a flame arrestor as well as spring-loaded lids on the filling and pouring spouts to avoid spilling if dropped. The flame-arrestor screen’s double-perforated metal surface stops flames from reaching the container. Both dispensing goods and collecting rubbish are possible with safety cans. Modifications to safety cans are not permitted. Many store-bought portable gasoline containers do not fulfill safety can requirements.
Storage in the Refrigerator
Domestic refrigerators, which are commercially accessible, have built-in ignition sources and should not be utilized to store flammable materials or explosive substances. All ignition sources include light bulbs, switches, temperature controls, standard outlets, motor-starting relays, thermal-overload devices, and heating strips (for frost control).
Refrigerators properly constructed and certified for storing volatile liquids or explosives should be used by everyone who requires one. Refrigerators and freezers that have been constructed or modified to properly hold flammable and/or combustible substances must be labeled as such.
How can flammable or combustible liquids provide a risk of fire or explosion?
Flammable liquids may give off enough vapor to generate burnable mixes with air at typical room temperatures. As a consequence, they may provide a significant fire risk. Flammable liquid flames spread quickly. They also produce a lot of heat and heavy, black, poisonous smoke clouds.
At temperatures over their flashpoint, combustible liquids release enough vapor to produce burnable mixes with the air. Hot combustible liquids may be just as dangerous as flammable liquids in terms of causing a fire.
If an ignition source is available, spray mists of flammable and combustible liquids in the air may burn at any temperature. Normally, the fumes of flammable and combustible liquids are not visible. Unless proper devices are utilized, they may be difficult to detect.
The majority of flammable and combustible liquids are easy to flow. A tiny spill on the workstation or the floor might quickly cover a wide area. Burning liquids may spread fire by flowing under doors, down staircases, and even into neighboring buildings. flammable or combustible liquids may readily be absorbed by materials like wood, paper, and fabric. Even after a spill is cleaned up, a deadly quantity of liquid may persist in nearby objects or garments, emitting harmful vapors.
Frequently Asked Questions(FAQs), “What is the difference between flammable and combustible?”
What is a flammable and combustible material?
A combustible substance is one that can burn (or combust) in the presence of air. If a combustible substance ignites quickly at room temperature, it is flammable. In other words, a combustible substance takes some effort to burn, but a flammable material ignites instantly when exposed to flame.
Is gas flammable or combustible?
For its low flashpoint and high vapor density, gasoline is classified as flammable. Because their Flashpoint is larger than 100 degrees F, oil and diesel fuel are classified as combustible. Gasoline emits flammable fumes that are three to four times heavier than air and may travel considerable distances on the ground.
What determines if a liquid is flammable or combustible?
The flashpoint of a substance determines its flammability. The minimal temperature at which a liquid creates a vapor above its surface in high quantity to ignite it is known as the flashpoint. The flashpoint of flammable liquids is less than 100°F. Lower flashpoints make it simpler to ignite liquids.
What is combustible material?
When exposed to fire or heat, combustible material will ignite, burn, assist the combustion, or emit flammable gases in the form in which it is utilized and in the circumstances expected. Combustible materials include wood, cardboard, rubber, and plastics.
Is alcohol flammable or combustible?
It is also known as alcohol and is flammable. Ethanol (C2H5OH) is a flammable, colorless, and mildly poisonous chemical substance that is commonly referred to simply as alcohol. Ethanol combustion produces carbon dioxide and water. Ethanol is a flammable and poisonous substance.
Can you ignite air?
Air will never spontaneously combust, and it cannot be forced to do so. The majority of air contains nitrogen, which is not combustible. Because nitrogen is non-reactive in general, it does not assist the combustion of other substances.
What makes a burn flammable?
If a flammable material is exposed to enough heat, it will burn. The auto-ignition temperature is the minimum temperature at which a flammable and combustible liquid can ignite — and continue to burn — without the presence of a spark or flame. | https://tamingfires.com/what-is-the-difference-between-flammable-and-combustible/ |
【Appearance】Clear, colorless liquid.
【Solubility in water】Miscible
【Melting Point】-73℃
【Boiling Point】140℃
【Vapor Pressure】6 (25℃)
【Density】0.928 g/cm3 (20℃)
【pKa/pKb】5.74 (pKb)
【Partition Coefficient】.06
【Heat Of Vaporization】37.7 kJ/mol
【Vapor Density】4.12
【Refractive Index】1.4115 (20℃)
First Aid Measures
【Ingestion】Never give anything by mouth to an unconscious person. Get medical aid immediately. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
【Inhalation】Get medical aid immediately. Remove from exposure to fresh air immediately. If breathing is difficult, give oxygen. Do not use mouth-to-mouth respiration if victim ingested or inhaled the substance; induce artificial respiration with the aid of a pocket mask equipped with a one-way valve or other proper respiratory medical device.
【Skin】Get medical aid. Flush skin with plenty of soap and water for at least 15 minutes while removing contaminated clothing and shoes. Remove contaminated clothing and shoes.
【Eyes】Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid immediately. Do NOT allow victim to rub or keep eyes closed.
Handling and Storage
【Storage】Keep away from heat, sparks, and flame. Keep away from sources of ignition. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances. Flammables-area.
【Handling】Remove contaminated clothing and wash before reuse. Use only in a well ventilated area. Ground and bond containers when transferring material. Use spark-proof tools and explosion proof equipment. Avoid contact with eyes, skin, and clothing. Empty containers retain product residue, (liquid and/or vapor), and can be dangerous. Avoid contact with heat, sparks and flame. Avoid ingestion and inhalation. Do not pressurize, cut, weld, braze, solder, drill, grind, or expose empty containers to heat, sparks or flames.
Hazards Identification
【Inhalation】Causes respiratory tract irritation. Vapors may cause dizziness or suffocation. Can produce delayed pulmonary edema. May cause burning sensation in the chest.
【Skin】Causes skin irritation. May cause dermatitis. May cause cyanosis of the extremities.
【Eyes】Produces irritation, characterized by a burning sensation, redness, tearing, inflammation, and possible corneal injury. Lachrymator. May cause chemical conjunctivitis and corneal damage.
【Ingestion】May cause gastrointestinal irritation with nausea, vomiting and diarrhea. Ingestion of large amounts may cause CNS depression.
【Hazards】Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will sp along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers.
【EC Risk Phrase】R 10 36/37/38
【EC Safety Phrase】S 16 26 36/37/39
【UN (DOT)】1993
Exposure Controls/Personal Protection
【Personal Protection】Eyes: Wear appropriate protective eyeglasses or chemical safety ggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166. Skin: Wear appropriate protective gloves to prevent skin exposure. Clothing: Wear appropriate protective clothing to prevent skin exposure.
【Respirators】A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant a respirator's use.
【Exposure Effects】Effects may be delayed.
Fire Fighting Measures
【Flash Point】29℃
【Autoignition】260℃
【Fire Fighting】Wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Will burn if involved in a fire. Use water spray to keep fire-exposed containers cool. Containers may explode in the heat of a fire. Extinguishing media: Use water spray to cool fire-exposed containers. Water may be ineffective. Do NOT use straight streams of water. In case of fire use water spray, dry chemical, carbon dioxide, or appropriate foam.
【Fire Potential】HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames.
Accidental Release Measures
【Small spills/leaks】Absorb spill with inert material, (e.g., dry sand or earth), then place into a chemical waste container. Avoid runoff into storm sewers and ditches which lead to waterways. Remove all sources of ignition. Use a spark-proof tool. Provide ventilation. A vapor suppressing foam may be used to reduce vapors.
Stability and Reactivity
【Disposal Code】1
【Incompatibilities】Oxidizing agents, acids.
【Stability】Stable at room temperature in closed containers under normal storage and handling conditions.
【Decomposition】Nitrogen oxides, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, nitrogen.
【Combustion Products】Fire may produce irritating, corrosive and/or toxic gases. | http://www.ts007.com/Chemical/122-07-6/msds |
Prudent Practices in the Laboratory : Handling and Disposal of Chemicals.
- Publication:
- Washington : National Academies Press, 1995.
- Format/Description:
- Book
1 online resource (443 pages)
- Status/Location:
-
Loading...
Options
|Location||Notes||Your Loan Policy|
Details
- Other records:
-
- Subjects:
- Hazardous substances.
Chemicals -- Safety measures.
Hazardous wastes.
- Form/Genre:
- Electronic books.
- Contents:
- Prudent Practices in the Laboratory
Copyright
PREFACE
ACKNOWLEDGMENTS
Contents
OVERVIEW AND RECOMMENDATIONS
THE CULTURE OF LABORATORY SAFETY (CHAPTER 1)
Education for Safety and Pollution Control
Factors Affecting Safety Practices in Laboratories
PRUDENT PLANNING OF EXPERIMENTS (CHAPTER 2)
EVALUATING HAZARDS AND ASSESSING RISKS IN THE LABORATORY (CHAPTER 3)
MANAGEMENT OF CHEMICALS (CHAPTER 4)
WORKING WITH CHEMICALS (CHAPTER 5)
WORKING WITH LABORATORY EQUIPMENT (CHAPTER 6)
DISPOSAL OF WASTE (CHAPTER 7)
Hazardous Chemical Waste
Multihazardous Waste
Chemical-Radioactive Waste
Chemical-Biological Waste
Radioactive-Biological Waste
Chemical-Radioactive-Biological Waste
LABORATORY FACILITIES (CHAPTER 8)
GOVERNMENTAL REGULATION OF LABORATORIES (CHAPTER 9)
RECOMMENDATIONS
Recommendations to the Environmental Protection Agency and Other Regulatory Agencies
Recommendations to the Industrial Sector
Recommendations to Chemical Laboratories
1 THE CULTURE OF LABORATORY SAFETY
1.A INTRODUCTION
1.B THE NEW CULTURE OF LABORATORY SAFETY
1.C RESPONSIBILITY AND ACCOUNTABILITY FOR LABORATORY SAFETY
1.D SPECIAL SAFETY CONSIDERATIONS IN ACADEMIC LABORATORIES
1.D.1 High School Teaching Laboratories
1.D.2 Undergraduate Teaching Laboratories
1.D.3 Academic Research Laboratories
1.E THE SAFETY CULTURE IN INDUSTRY
1.F FACTORS THAT ARE CHANGING THE CULTURE OF SAFETY
1.F.1 Advances in Technology
1.F.2 The Culture of Pollution Prevention
1.F.2.1 Waste Management Hierarchy for Pollution Prevention
1.F.2.2 Making Pollution Prevention Work
1.F.3 Changes in the Legal and Regulatory Climate
1.G ORGANIZATION OF THIS BOOK
2 PRUDENT PLANNING OF EXPERIMENTS
2.A INTRODUCTION
2.B LEVELS OF FORMALITY IN EXPERIMENT PLANNING.
2.C INDIVIDUAL RESPONSIBILITIES FOR PLANNING EXPERIMENTS
2.D INSTITUTIONAL POLICIES AND EMERGENCY RESPONSE PLANNING
2.E STEPS FOR PLANNING AN EXPERIMENT
2.E.1 Chapter 3: Evaluating Hazards and Assessing Risks in the Laboratory
2.E.2 Chapter 4: Management of Chemicals
2.E.3 Chapter 5: Working with Chemicals
2.E.4 Chapter 6: Working with Laboratory Equipment
2.E.5 Chapter 7: Disposal of Waste
2.E.6 Chapter 8: Laboratory Facilities
2.E.7 Chapter 9: Governmental Regulation of Laboratories
3 EVALUATING HAZARDS AND ASSESSING RISKS IN THE LABORATORY
3.A INTRODUCTION
3.B SOURCES OF INFORMATION
3.B.1 Chemical Hygiene Plan
3.B.2 Material Safety Data Sheets
3.B.3 Laboratory Chemical Safety Summaries
3.B.4 Labels
3.B.5 Additional Sources of Information
3.B.6 Computer Services
3.B.6.1 The National Library of Medicine Databases
3.B.6.2 Chemical Abstracts Databases
3.B.6.3 Informal Forum
3.C TOXIC EFFECTS OF LABORATORY CHEMICALS
3.C.1 Basic Principles
3.C.1.1 Dose-Response Relationships
3.C.1.2 Duration and Frequency of Exposure
3.C.1.3 Routes of Exposure
3.C.1.3.1 Inhalation
3.C.1.3.2 Contact with Skin or Eyes
3.C.1.3.3 Ingestion
3.C.1.3.4 Injection
3.C.2 Types of Toxins
3.C.2.1 Irritants
3.C.2.2 Corrosive Substances
3.C.2.3 Allergens
3.C.2.4 Asphyxiants
3.C.2.5 Carcinogens
3.C.2.6 Reproductive and Developmental Toxins
3.C.2.7 Neurotoxins
3.C.2.8 Toxins Affecting Other Organs
3.C.3 Assessing Risks Due to the Toxic Effects of Laboratory Chemicals
3.C.3.1 Acute Toxicants
3.C.3.2 Corrosive Substances, Irritants, and Allergens
3.C.3.3 Carcinogens
3.C.3.4 Reproductive and Developmental Toxins
3.D FLAMMABLE, REACTIVE, AND EXPLOSIVE HAZARDS
3.D.1 Flammable Hazards
3.D.1.1 Flammable Substances
3.D.1.2 Flammability Characteristics.
3.D.1.2.1 Flash Point
3.D.1.2.2 Ignition Temperature
3.D.1.2.3 Limits of Flammability
3.D.1.3 Classes of Flammability
3.D.1.4 Causes of Ignition
3.D.1.4.1 Spontaneous Combustion
3.D.1.4.2 Ignition Sources
3.D.1.4.3 Oxidants Other Than Oxygen
3.D.1.5 Special Hazards
3.D.2 Reactive Hazards
3.D.2.1 Water Reactives
3.D.2.2 Pyrophorics
3.D.2.3 Incompatible Chemicals
3.D.3 Explosive Hazards
3.D.3.1 Explosives
3.D.3.2 Peroxides
3.D.3.3 Other Oxidizers
3.D.3.4 Dusts
3.D.3.5 Explosive Boiling
3.D.3.6 Other Considerations
3.D.4 The Dirty Dozen
3.E PHYSICAL HAZARDS
3.E.1 Compressed Gases
3.E.2 Nonflammable Cryogens
3.E.3 High-Pressure Reactions
3.E.4 Vacuum Work
3.E.5 Ultraviolet, Visible, and Near-Infrared Radiation
3.E.6 Radiofrequency and Microwave Hazards
3.E.7 Electrical Hazards
3.E.8 Magnetic Fields
3.E.9 Cuts, Slips, Trips, and Falls
3.F BIOHAZARDS
3.G HAZARDS FROM RADIOACTIVITY
4 MANAGEMENT OF CHEMICALS
4.A INTRODUCTION
4.B SOURCE REDUCTION
4.B.1 Importance of Minimizing Chemical Orders
4.B.2 Strategies to Minimize Hazardous Waste Generation
4.B.2.1 Microscale Work
4.B.2.2 Step-by-Step Planning for Minimization
4.B.2.3 Substitution of Materials
4.B.3 Strategies to Avoid Multihazardous Waste Generation
4.C ACQUISITION OF CHEMICALS
4.C.1 Ordering Chemicals
4.C.2 Receiving Chemicals
4.C.3 Responsibilities for Chemicals Being Shipped or Transported
4.D INVENTORY AND TRACKING OF CHEMICALS
4.D.1 General Considerations
4.D.2 Exchange or Transfer of Chemicals
4.D.3 Labeling Commercially Packaged Chemicals
4.D.4 Labeling Other Chemical Containers
4.D.5 Labeling Experimental Materials
4.D.6 Use of Inventory and Tracking Systems in Emergency Planning
4.E STORAGE OF CHEMICALS IN STOCKROOMS AND LABORATORIES.
4.E.1 General Considerations
4.E.2 Containers and Equipment
4.E.3 Storing Flammable and Combustible Liquids
4.E.4 Storing Gas Cylinders
4.E.5 Storing Highly Reactive Substances
4.E.6 Storing Toxic Substances
4.F RECYCLING OF CHEMICALS, CONTAINERS, AND PACKAGING
4.F.1 General Considerations
4.F.2 Solvent Recycling
4.F.3 Mercury Recycling
4.F.4 Reclamation of Heavy Metals
5 WORKING WITH CHEMICALS
5.A INTRODUCTION
5.B PRUDENT PLANNING
5.C GENERAL PROCEDURES FOR WORKING WITH HAZARDOUS CHEMICALS
5.C.1 Personal Behavior
5.C.2 Minimizing Exposure to Chemicals
5.C.2.1 Avoiding Eye Injury
5.C.2.2 Avoiding Ingestion of Hazardous Chemicals
5.C.2.3 Avoiding Inhalation of Hazardous Chemicals
5.C.2.4 Avoiding Injection of Hazardous Chemicals
5.C.2.5 Minimizing Skin Contact
5.C.2.6 Clothing and Protective Apparel
5.C.3 Housekeeping
5.C.4 Transport of Chemicals
5.C.5 Storage of Chemicals
5.C.6 Disposal of Chemicals
5.C.7 Use and Maintenance of Equipment and Glassware
5.C.8 Handling Flammable Substances
5.C.9 Working with Scaled-up Reactions
5.C.10 Responsibility for Unattended Experiments and Working Alone
5.C.11 Responding to Accidents and Emergencies
5.C.11.1 General Preparation for Emergencies
5.C.11.2 Handling the Accidental Release of Hazardous Substances
5.C.11.3 Notification of Personnel in the Area
5.C.11.4 Treatment of Injured and Contaminated Personnel
5.C.11.5 Spill Containment
5.C.11.6 Spill Cleanup
5.C.11.7 Handling Leaking Gas Cylinders
5.C.11.8 Handling Spills of Elemental Mercury
5.C.11.9 Responding to Fires
5.D WORKING WITH SUBSTANCES OF HIGH TOXICITY
5.D.1 Planning
5.D.2 Experiment Protocols Involving Highly Toxic Chemicals
5.D.3 Designated Areas
5.D.4 Access Control.
5.D.5 Special Precautions for Minimizing Exposure to Highly Toxic Chemicals
5.D.6 Preventing Accidents and Spills with Substances of High Toxicity
5.D.7 Storage and Waste Disposal
5.D.8 Multihazardous Materials
5.E WORKING WITH BIOHAZARDOUS AND RADIOACTIVE MATERIALS
5.E.1 Biohazardous Materials
5.E.2 Radioactive Materials
5.F WORKING WITH FLAMMABLE CHEMICALS
5.F.1 Flammable Materials
5.F.2 Flammable Liquids
5.F.3 Flammable Gases
5.F.4 Catalyst Ignition of Flammable Materials
5.G WORKING WITH HIGHLY REACTIVE OR EXPLOSIVE CHEMICALS
5.G.1 Overview
5.G.2 Reactive or Explosive Compounds
5.G.2.1 Personal Protective Apparel
5.G.2.2 Protective Devices
5.G.2.3 Evaluating Potentially Reactive Materials
5.G.2.4 Determining Reaction Quantities
5.G.2.5 Conducting Reaction Operations
5.G.3 Organic Peroxides
5.G.3.1 Peroxide Detection Tests
5.G.3.2 Disposal of Peroxides
5.G.4 Explosive Gases and Liquefied Gases
5.G.5 Hydrogenation Reactions
5.G.6 Reactive or Explosive Materials Requiring Special Attention
5.G.7 Chemical Hazards of Incompatible Chemicals
5.H WORKING WITH COMPRESSED GASES
5.H.1 Chemical Hazards of Compressed Gases
5.H.2 Specific Chemical Hazards of Select Gases
6 WORKING WITH LABORATORY EQUIPMENT
6.A INTRODUCTION
6.B WORKING WITH WATER-COOLED EQUIPMENT
6.C WORKING WITH ELECTRICALLY POWERED LABORATORY EQUIPMENT
6.C.1 General Principles
6.C.1.1 Outlet Receptacles
6.C.1.2 Wiring
6.C.1.3 General Precautions for Working with Electrical Equipment
6.C.1.4 Personal Safety Techniques for Use with Electrical Equipment
6.C.1.5 Additional Safety Techniques for Equipment Using High Current or High Voltage
6.C.2 Vacuum Pumps
6.C.3 Refrigerators and Freezers
6.C.4 Stirring and Mixing Devices
6.C.5 Heating Devices
6.C.5.1 Ovens.
6.C.5.2 Hot Plates.
- Notes:
- Description based on publisher supplied metadata and other sources.
- Local notes:
- Electronic reproduction. Ann Arbor, Michigan : ProQuest Ebook Central, 2021. Available via World Wide Web. Access may be limited to ProQuest Ebook Central affiliated libraries.
- Other format:
- Print version: Staff, National Research Council Prudent Practices in the Laboratory
- ISBN:
- 9780309560733
9780309052290
- OCLC: | https://franklin.library.upenn.edu/catalog/FRANKLIN_9977920950903681 |
24.0815 Storage—-General requirements.
Before being distributed or used, all imported hazardous materials shall be stored in a way that will protect against the unintentional release of the materials to the environment. Where storage is defined as 10% of the reportable quantity or 55 gallons or greater, protective measures shall include:
(1) Segregation of incompatible materials including segregation of all unregulated incompatible materials stored in the same area;
(2) Protection from exposure to weather through storage in an indoor area, including adequate roofs, walls, and floors to prevent rain from reaching PCB’s;
(3) Location in an area that if flooded would pose no risk to populated areas or the water supply;
(4) Protection from all sources of heat, fire hazards, and adequate ventilation;
(5) Bilingual warning signs, in both English and Samoan, indicating the type of substances stored and their hazards shall be posted outside the storage area;
(6) Adequate security, including fences, barriers or other means of preventing unauthorized access, and adequate lighting to promote discovery of spills at night and to prevent spills caused by vandalism;
(7) Containers used to store materials shall be in good condition, shall be compatible with the items being stored, and shall be closed at all times while in storage. Containers used to store materials shall be handled in a way that does not cause the containers to rupture or leak;
(8) Inspections. All storage areas shall be inspected weekly to detect leaking or deteriorating containers and to ensure that all emergency equipment is functioning. All leaking containers and their contents shall be transferred immediately to properly marked non-leaking containers, and spilled or leaked materials cleaned up immediately using absorbents or other adequate means. Inspections shall be conducted by facility personnel whose training has been documented and verified in compliance with sections 24.0811. Inspections shall be documented and records kept on the premises for at least one year.
(9) Labeling in accordance with requirements of USDOT regulations set forth in 49 CFR part 172, which are hereby incorporated by reference.
(10) Facility requirements as follows:
(A) All facilities where hazardous substances are accumulated or staged shall be off-limits to unauthorized personnel, and appropriate steps shall be taken to protect the public health and safety;
(B) All facilities where hazardous substances are accumulated or staged, where pollution control devices are operating, or where treatment facilities are located shall have the following devices and equipment for personnel protection:
(i) Eyewash station;
(ii) Shower;
(iii) Emergency communication equipment;
(iv) Fire protection as approved by the fire chief;
(v) Personal protection suits, gloves and boots;
(vi) Spill response equipment appropriate for the amounts and types of materials handled at the facility.
(C) Spill prevention equipment. All facilities that accumulate or stage material characterized as hazardous and having the potential to become hazardous waste if released into the environment shall have appropriate containment and spill-prevention controls for preventing a release. Containment devices shall be capable of holding the content of the largest container or 10 percent of all material accumulated or staged in the area, whichever is greater. Such facilities shall be equipped with weather-protection devices that are sufficient for preventing rain or runoff from entering the facility.
(11) A sign or signs bearing the legend “Danger: Unauthorized Personnel Keep Out” shall be posted at each entrance or active part of a facility and at other locations in numbers sufficient to be seen from any approach. The legend shall be written in English and Samoan and shall be legible from a distance of at least 50 feet. Signs in Samoan and English warning of hazards (e.g., “No Smoking”) also shall be posted.History: Rule 02-01, eff 30 Aug 01. | https://new.asbar.org/regulation/24-0815-storage-general-requirements/ |
Workplace fire safety
By following these simple steps you can reduce the likelihood of adding to the UAE's fire statistics.
By following these simple steps you can reduce the likelihood of adding to the statistics, but if fire does occur, keep the impact to the absolute minimum. Glen Sumber, senior fire consultant and Dean McGrail, director, WSP Fire, explain.
Fires in the workplace are all too common and have resulted in the loss to many businesses, both in the UAE and internationally.
Loss from a fire can be measured in physical injury to yourself, employees and anyone visiting or near the premises; in damage to the premises and its contents and financially through loss of business, reputation and on occasion through court action.
But statistics show that by taking some simple steps, the majority could be prevented from happening or at least reduce the consequences.
A relatively small fire, lasting no longer than the average lunch break could easily cause the type of injury and damage that may leave long term physical effects and the closure of a business. So what can facilities managers do to reduce the risk of fire and minimise losses?
There are a number of simple steps FMs can take to reduce the risk of fire occurring in the workplace and any subsequent effects on you, the employees and the business.
Instruction
Make sure all employees receive suitable fire training on a regular basis. The training should include:
1. A simple explanation of what fire is, how it occurs and the likely causes;
2. Fire reduction methods all employees can take;
3. The emergency plan - how you are notified of a fire in the office, what to do if you hear the alarm and how to contact the emergency services;
4. Basic fire fighting methods;
5. The escape routes out of the building and the location of the assembly point;
6. What to do after a fire has occurred, including any business recovery plans;
7. Specialist training where required, e.g. fire wardens and evacuation marshalling.
Escape routes
In the event of a fire, it is the escape routes that will get everyone in the workplace out of the building in the shortest possible time.
For this reason they must remain usable at all times.
Remember to consider:
1. A general rule of thumb says that escape routes should be a minimum of 1.1 metres wide;
2. Escape routes should not be used as storage areas. Small levels of storage can take place but items must be in a fire proof cabinet which must not obstruct the route;
3. Ensure escape doors are working correctly - the automatic closer operates, double doors align with each other when closed and smoke seals are not damaged;
4. Do not fix items to escape doors that have not been approved by the manufacturer - you will affect its performance in a fire;
5. Implement a system for checking the escape routes daily and one that controls changes to the routes either due to construction or maintenance;
6. Ensure escape signage is clearly visible from all locations in your workplace;
7. Affix escape notices adjacent to final escape doors.
Housekeeping
Poor housekeeping creates the right environment for a fire to take place, providing both a place where ignition can occur together with a ready source of fuel.
It may also create obstructions to the escape routes.
That said, it is perhaps one of the easiest risks to control:
1. Ensure outer clothing is hung up and not allowed to become piled in an escape route or on a heater;
2. In an office, a clear desk policy will help keep paper and other flammable materials to a minimum;
3. Use metal bins and ensure employees do not allow waste to build up on or near their work areas;
4. Waste storage should be in a dedicated area, away from escape routes and regularly emptied;
5. Take care when disposing of oily rags as they can self-ignite;
6. Set a good housekeeping example - if you keep your area clear other workers will be more inclined to follow suit.
Storage
Most workplaces have storage and it is likely that the stored items will be flammable.
This is particularly the case where large quantities of paper records are stored, especially if they are not kept in a metal cabinet.
Storage of the more risky, highly flammable substances such as solvents and gasses, should be kept to an absolute minimum.
If incorrectly stored even small quantities of a highly flammable substance may greatly enhance the size of fire and any effects.
Storage considerations should therefore include:
1. Store sensibly and consider adjacent heat sources - large quantities of photocopier paper stored behind the copier provide a fuel for the heat from the copier to ignite;
2. Only store sufficient quantities of substances for your immediate needs - consider off-site storage for items that are not required daily;
3. Remove highly flammable substances altogether or where this is not possible reduce the amount stored in the workplace.
Follow the more stringent legislation (like the Dangerous Substances and Explosive Atmospheres Regulations 2002), to control highly flammable substances;
4. No naked flames adjacent to storage areas containing flammable substances.
Electrical safety
The majority of fires in workplaces are caused by faulty electrical appliances and leads (Fire Statistics Monitor. Issues 01/08, 02 May 2008 - Communities and Local Government).
Steps that can be taken to reduce the risk of fire will include:
1. Ensuring all electrical equipment is not damaged and working correctly - look for fraying cables, exposed electrical connections and cracked and ‘sooty' marking on casings;
2. All fixed electrical equipment, e.g. photocopiers, should be maintained in accordance with the manufacturer's guidelines or industry standards.
This should be carried out by a competent person;
3. Portable electrical equipment, e.g. drills, should be tested at a period suitable for the equipment.
This may take the form of a portable appliance test (PAT), but may also be achieved through inspection by a competent electrical engineer;
4. Check the equipment and ensure the correct fuse has been installed. If you do not feel confident doing this, get a competent electrical engineer to help;
5. Do not overload sockets. Multi-point plug adaptors can overheat and create a source of ignition. Likewise, multi-point extension leads can also become overloaded and catch fire;
6. Do not store flammable materials against items of electrical equipment.
The equipment may not catch fire but the heat generated may create the ignition source for the stored flammable materials to ignite.
Gases and flammable liquids
Gases and flammable liquids in workplaces will probably be used and stored in small quantities. But bulk purchasing of substances, aerosols and solvents, for example, may create a larger risk.
Steps to be implemented to ensure safe use and storage include:
1. Store and use only sufficient quantities for day to day activities;
2. Keep all gases and flammable liquids in a proprietary metal cabinet with suitable hazard labels clearly placed on the front;
3. Ensure all gases and flammable liquids are used for the purpose they are designed;
4. Leaks and spillages should be cleaned up immediately;
5. Ensure there are no naked flames in the vicinity of stored substances or where they are being used.
Fire safety
Poorly maintained or inoperative fire safety equipment, such as fire detection and alarm systems and emergency lighting installations, could put employees in danger if a fire were to occur.
They are designed to both detect a fire in its early stages and provide adequate warning to all occupants so that safe escape can be achieved.
Maintenance requirements are stipulated by both the US and European standards (the fire and life safety codes adopted in the UAE) and should be undertaken by suitably qualified persons.
The person in control of the workplace should assure themselves that all such systems are fully working and maintained and tested in accordance with the relevant standard.
Considerations to follow to ensure systems operate correctly include:
1. Know and understand the systems your building has;
2. Employ a competent contractor to undertake all maintenance and testing of fire safety systems;
3. Regularly test the fire alarms to ensure they work and employees know where to go;
4. Request confirmation of all work undertaken and keep copies of the test certificates;
5. Do not cover or paint-over smoke detectors;
6. Ensure manual call points (break glass units) are accessible at all times;
7. Portable fire extinguishers should be maintained at least once each year;
8. Check the main fire alarm panel for fault indicator lights. The panel will usually be located in the main entrance to the building.
If a fault light is indicated contact your maintenance contractor.
General maintenance activities
Maintenance activities can also be the cause of a fire in your workplace.
Many maintenance activities create heat, sparks or use naked flames. Activities may also impact the fire safety systems or obstruct escape routes.
It is imperative that suitable controls are implemented that should consider:
1. A check and approval system for all maintenance activities in your workplace so all risks can be assessed before work starts;
2. Permits to work such as for hot working, isolation of fire systems and interruption to sprinkler supplies;
3. Ensuring the maintenance workers fully understand how they will interface with the rest of the building and the employees;
4. Where an isolation or interruption to a fire safety system occurs, ensure suitable alternative measures are implemented;
5. Ensure additional portable fire extinguishers are provided within the maintenance site.
Arson
Whilst arson is uncommon in the UAE at present, to reduce its possibility the following measures should be considered:
1. Ensure the workplace is secure at all times - remove areas where trespassers can gain access unobserved and keep fences maintained;
2. External bins should be kept away from the building - at least six metres;
3. Install security cameras in prominent locations - they double as a visible deterrent;
4. Investigate frequent, small fires within the workplace considering the possibility of in-house arson;
5. Ensure all doors are kept shut and locked (but remember if they are an escape door they must be easily opened from the direction of escape);
6. Check all doors are firmly locked shut when your employees leave at night.
Use of unauthorised equipment
Equipment that has been brought in from home or elsewhere may not have received the same level of maintenance and testing as your equipment.
It may also be unsuitable for the particular work environments at your workplace.
1. Do not allow equipment to be brought in and used without first ensuring it is maintained and fit for purpose in the environment;
2. Unwanted (false) alarms where no cause can be found, may sometimes be the result of home equipment such as toasters;
3. Canvass your employees to ascertain what equipment they require - this may prevent equipment being brought in. | https://www.constructionweekonline.com/article-3301-workplace-fire-safety |
N-Benzyl-4-Piperidone Formal Name:
1-(phenylmethyl)-4-piperidinone
CAS Number:3612-20-2
Synonyms: 1-Benzyl-4-piperidone:
NSC 77933
Molecular Formula: C12H15NO
Formula Weight: 189.3
Purity: ≥98%
4-Piperidone Monohydrate usage: Pharmaceutical Intermediates
Appearance & Physical State: clear to yellow oilyliquid
Density: 1.069
Boiling Point: 114-116ºC (0.4 mmHg)
Flash Point: 71ºC
Refractive Index: 1.539-1.54
Water Solubility: 12 g/L (20 ºC)
Stability: Stable under normal temperatures and pressures.
Storage Condition: Keep away from heat, sparks, and flame. Keep away from sources of ignition. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances
Factory showing
Visiting Factory
Exhibition
High Purity Piperidone CAS NO.3612-20-2 chemical name 1-Benzyl-4-piperidone date analysis
Our products are divided into three parts : food &Feed Additives,pharmaceutical intermediates and oilfield auxiliaries. the feed additives devotes to the research and production of Betaine Hcl, Anhydrous Betaine , Compound Betaine,DMT, DMPT , tRIMETHYLAMINE n-OXIDE( Tmao ) , And on going technology updates , the technology in a leading position . Pharmaceutical intermediates and oilfield anuxiliaries include piperidone series , anthracene series, glycerol derivatives. | https://www.sdefine.com/piperidone/54706295.html |
Homes and offices alike typically hold a number of chemical products. This could range from cleaning and maintenance supplies, to bathroom products, medicines, renovation materials, and more. Most people tend to just stick the various bottles in a closet or cabinet and forget about them. However, if chemical products are not stored safely, they could leak, cause corrosion, and even explode. It is even more dangerous if there are small children or pets around. Since most chemical products are fatally toxic, they could cause severe illness and death if consumed. This guide will help you learn how to store chemical products correctly so that they do not cause any harm.
Before starting to store chemical products correctly, it is necessary to identify all of them. For this, you would need to go through each room and make a list of all the chemical products that are kept there. Check their labels to find out what hazards they pose. The most common types are corrosive, flammable, explosive (or reactive), and toxic. By knowing the potential hazards, it is easier to group, sort, and store the products.
- Make sure that all products are in their original containers, and that the container is still in good condition. It should not be rusty, broken, cracked, or leaking.
- All products should still have the original labels on them. If the label is worn away, create a new label, with the hazard information or symbols on it. If the label has disappeared entirely, it is best to discard the product to avoid future confusion.
- If you have children or pets at home, always keep chemical products locked or securely out of their reach.
- When arranging and storing chemical products, use a pair of durable gloves in case any containers are leaking.
- In general, group like products together. It helps further to use a storage bin so that they do not fall over easily.
- For flammable materials, find a safe, cool place to store them. This should ideally be away from any heat sources and out of direct sunlight as well.
- Medicinal products should always bear their original labels or the prescription. Never empty medicines into other bottles.
- Pressurized containers should not be left in areas that are prone to dampness, dripping or leaking water, or any other type of wetness. This could cause the containers to rust, and leak the contents.
- For liquid chemical products, it is a good idea to keep a spare bag of sand or kitty litter close by. If there is ever a leak, simply spread the sand or cat litter on the floor to soak up the chemical leak as quickly as possible.
- Keep fuels and additional flammable materials, such as paint thinner, in the shed or the garage. They should always be stored in proper containers that are meant to hold such materials. Make sure that the garage has sufficient ventilation.
- Pesticides are best stored in the garage as well. However, if you live in a cold area, do be aware that very cold temperatures cause a separation of materials in the pesticide. Only buy and store the amounts that you will be using.
- Do a general check of your chemical products at least once per year. In this way, you can get rid of any old or unwanted items, and check that other containers are still in good condition.
- If you keep chemical products on a shelving unit, first make sure that it is solid. It should not be unstable or in weak condition.
Additional Resources
- Chemical Storage Tips (PDF) – Follow these guidelines for additional tips on how to safely store chemicals to avoid any hazards.
- Household Chemicals – Be aware of the surprising number of household products that are flammable, toxic, explosive, or dangerous in other ways.
- Chemical handling practices when dealing with chemicals can keep you and your family safe.
- Storing Chemicals the Right Way – Chemicals should always be labeled correctly and kept in original containers.
- Swimming Pool Chemicals (PDF) – Home pool owners should be careful to keep pool maintenance products completely dry.
- Buy Storage Buildings (PDF) – Home pool owners should be careful to keep pool maintenance products completely dry.
- Keeping and Disposing of Chemicals (PDF) – Watch a slideshow on the proper procedures for storing and discarding chemicals.
- Storing Flammable Chemicals – Review important information on flammable chemical hazard classifications and how to store them appropriately.
- Hazardous Materials – Find out how to sort hazardous materials and then store them in the safest way possible. | https://alansfactoryoutlet.com/storage-buildings-chemical-storage-safety-tips/ |
Working with Flammables and Reactives in the Laboratory
Course Description:
This course covers lab safety as it relates to the safe handling of flammables and reactives in the lab. Workplace fires and explosions are not only common—but also deadly. By virtue of the type of work conducted in laboratories—including working with hazardous chemicals, mixing chemicals, high heat, and intense pressure—the risk of fire or explosion can be high. It is essential that lab workers know how to identify these hazards; how to properly handle flammable, reactive, and combustible materials to avoid fire and explosions; and how to respond should one occur.
By the end of this session, trainees will be able to:
- Identify flammable and reactive hazards in the laboratory;
- Define flammables, reactives, and combustibles;
- Outline safe handling and storage for these materials;
- Discuss methods of prevention; and
- Discuss emergency response measures, safety equipment, and evacuation procedures.
Why “Working with Flammables and Reactives in the Laboratory” Matters:
There are a variety of regulations and guidelines that govern the use, handling, and storage of flammable and reactive materials in the lab. Some are specific to the laboratory and others are not, but all are relevant to lab safety regarding fires and explosions.
- Emergency action plans and fire prevention plans are required by federal rules under 29 CFR 1910.33 to 1910.39.
- OSHA’s Flammable and Combustible Liquids Standard (29 CFR 1910.106) explains what these substances are, how to store and use them safely, and how to prevent contact between these liquids and ignition sources.
Key Points:
The key points from this course include:
- Most chemicals in the lab are flammable and/or reactive. Follow labels, but when in doubt, assume flammability.
- Always store and handle flammables and reactives according to guidelines.
- Common sense and good housekeeping can avoid triggers for fires and explosions. A clean, organized lab goes a long way toward reducing hazards.
- Quick response and knowledge of procedures are critical in an emergency. Know your lab and the chemicals being used there. Test yourself on knowing what the signs and labels mean. Practice on emergency equipment and participate in drills.
- No Smoking! This is the number one rule to follow around flammables and reactives. | https://simplifytraining.com/course/working-with-flammables-and-reactives-in-the-laboratory/ |
Distance Units:
Distance Units
Standard
Metric
Skip Data Menu
Key Info
Identification
Protective Equipment / Clothing
Protective Distance
Fire Fighting Procedures
Reactivities / Incompatibilities
Treatment Overview
Basic
CAS Registry Number
EPA Hazardous Waste No.
Major Uses
Molecular Formula
Shipment Methods / Regulations
Shipping Name / Number
Storage Conditions
Substance Categories
Synonyms
Properties
Property Summary
Boiling Point
Color / Form
Density / Specific Gravity
Flash Point
Ionization Potential
Melting Point
Molecular Formula
Molecular Weight
Odor
Odor Threshold
Other Properties
Polymerization
Solubilities
Vapor Pressure
Viscosity
Hazmat
DOT Emergency Guidelines
Protective Distance
NFPA Hazard Classification
GHS Classification
Chemical Reactivity
Fire Potential
Fire Fighting Procedures
Protective Equip. / Clothing
Explosive Limits / Potential
Reactivities / Incompatibilities
Other Fire Fighting Hazards
Cleanup Methods
Disposal Methods
Medical
Treatment Overview
Health Effects
AEGL
Threshold Limit Values
NIOSH Recommendations
OSHA Standards
Skin / Eye / Resp. Irritants
Other Preventive Measures
Range of Toxicity
Laboratory
Environment
Exposure Summary
Environmental Fate
CERCLA Quantities
Non-Human Toxicity Values
Ecotoxicity Values
Soil Adsorption / Mobility
Volatilization
Propargyl Alcohol
CAS RN:
107-19-7
DOT Emergency Guidelines
GUIDE 131
F
LAMMABLE LIQUIDS - TOXIC
POTENTIAL HAZARDS
HEALTH
· TOXIC; may be fatal if inhaled, ingested or absorbed through skin.
· Inhalation or contact with some of these materials will irritate or burn skin and eyes.
· Fire will produce irritating, corrosive and/or toxic gases.
· Vapors may cause dizziness or suffocation.
· Runoff from fire control or dilution water may cause pollution.
FIRE OR EXPLOSION
· HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames.
· Vapors may form explosive mixtures with air.
· Vapors may travel to source of ignition and flash back.
· Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks).
· Vapor explosion and poison hazard indoors, outdoors or in sewers.
· Those substances designated with a
(P)
may polymerize explosively when heated or involved in a fire.
· Runoff to sewer may create fire or explosion hazard.
· Containers may explode when heated.
· Many liquids are lighter than water.
PUBLIC SAFETY
· CALL EMERGENCY RESPONSE Telephone Number on Shipping Paper first. If Shipping Paper not available or no answer, refer to appropriate telephone number listed on the inside back cover.
· As an immediate precautionary measure, isolate spill or leak area for at least 50 meters (150 feet) in all directions.
· Keep unauthorized personnel away.
· Stay upwind, uphill and/or upstream.
· Ventilate closed spaces before entering.
PROTECTIVE CLOTHING
· Wear positive pressure self-contained breathing apparatus (SCBA).
· Wear chemical protective clothing that is specifically recommended by the manufacturer. It may provide little or no thermal protection.
· Structural firefighters' protective clothing provides limited protection in fire situations ONLY; it is not effective in spill situations where direct contact with the substance is possible.
EVACUATION
Spill
· See the
Initial Isolation and Protective Action Distances
for highlighted materials. For non-highlighted materials, increase, in the downwind direction, as necessary, the isolation distance shown under "
PUBLIC SAFETY
".
Fire
· If tank, rail car or tank truck is involved in a fire, ISOLATE for 800 meters (1/2 mile) in all directions; also, consider initial evacuation for 800 meters (1/2 mile) in all directions.
In Canada, an Emergency Response Assistance Plan (ERAP) may be required for this product. Please consult the shipping document and/or the Canada and United States National Response Centers reference document.
EMERGENCY RESPONSE
FIRE
CAUTION: All these products have a very low flash point: Use of water spray when fighting fire may be inefficient.
Small Fire
· Dry chemical, CO
2
, water spray or alcohol-resistant foam.
Large Fire
· Water spray, fog or alcohol-resistant foam.
· Move containers from fire area if you can do it without risk.
· Dike fire-control water for later disposal; do not scatter the material.
· Use water spray or fog; do not use straight streams.
Fire involving Tanks or Car/Trailer Loads
· Fight fire from maximum distance or use unmanned hose holders or monitor nozzles.
· Cool containers with flooding quantities of water until well after fire is out.
· Withdraw immediately in case of rising sound from venting safety devices or discoloration of tank.
· ALWAYS stay away from tanks engulfed in fire.
· For massive fire, use unmanned hose holders or monitor nozzles; if this is impossible, withdraw from area and let fire burn.
SPILL OR LEAK
· Fully encapsulating, vapor-protective clothing should be worn for spills and leaks with no fire.
· ELIMINATE all ignition sources (no smoking, flares, sparks or flames in immediate area).
· All equipment used when handling the product must be grounded.
· Do not touch or walk through spilled material.
· Stop leak if you can do it without risk.
· Prevent entry into waterways, sewers, basements or confined areas.
· A vapor-suppressing foam may be used to reduce vapors.
Small Spill
· Absorb with earth, sand or other non-combustible material and transfer to containers for later disposal.
· Use clean, non-sparking tools to collect absorbed material.
Large Spill
· Dike far ahead of liquid spill for later disposal.
· Water spray may reduce vapor, but may not prevent ignition in closed spaces.
FIRST AID
· Ensure that medical personnel are aware of the material(s) involved and take precautions to protect themselves.
· Move victim to fresh air.
· Call 911 or emergency medical service.
· Give artificial respiration if victim is not breathing.
· Do not use mouth-to-mouth method if victim ingested or inhaled the substance; give artificial respiration with the aid of a pocket mask equipped with a one-way valve or other proper respiratory medical device.
· Administer oxygen if breathing is difficult.
· Remove and isolate contaminated clothing and shoes.
· In case of contact with substance, immediately flush skin or eyes with running water for at least 20 minutes.
· Wash skin with soap and water.
· In case of burns, immediately cool affected skin for as long as possible with cold water. Do not remove clothing if adhering to skin.
· Keep victim calm and warm.
· Effects of exposure (inhalation, ingestion or skin contact) to substance may be delayed.
ERG 2016
Find more information on this substance at:
Hazardous Substances Data Bank
,
TOXNET
,
PubMed
PDF documents can be viewed with the free
Adobe® Reader
Copyright
,
Privacy
,
Accessibility
U.S. National Library of Medicine
, 8600 Rockville Pike, Bethesda, MD 20894
National Institutes of Health
, | https://webwiser.nlm.nih.gov/getSubstanceData.do?substanceId=146&displaySubstanceName=1-Propyne-3-ol&STCCID=&UNNAID=&selectedDataMenuItemID=46&catId=54 |
For optimum performance, mix Borchi® A-111 EU into resin before the addition of other formulation components. This process prevents bubble formation during compounding and application processes. 0.1 – 0.5 % additive by weight (as supplied) based on total formulation.
Storage
Please consult Borchi® A-111 EU product label for storage and handling instructions.
Store locked up. Keep away from heat, sparks and open flame. Prevent electrostatic charge build-up by using common bonding and grounding techniques. Store in a cool, dry place out of direct sunlight. Store in tightly closed container. Store in a well-ventilated place. Keep in an area equipped with sprinklers. Store away from incompatible materials (see Section 10 of the SDS).
Safety
Please refer to our safety data sheet for information relating to product safety
SAFETY DATA SHEET
- Availability : Americas , Asia , EMEA
- Media : Solvent-based
Registration : | https://www.borchers.com/product/borchi-a-111-eu/ |
Mixing incompatible materials together can result in explosions, fires and severe injuries. An article in Lab Manager Magazine reports that improper storage of chemicals accounts for 25 percent of all chemical accidents in labs. The article describes the following examples of two serious lab incidents, both involving nitric acid:
- Nitric acid mixed with an organic solvent exploded in a university lab.
- Someone put nitric acid into a container previously used for methanol. Over the next 12 to 16 hours, the nitric acid reacted with the residual methanol. This created carbon dioxide and caused pressure to build up. Eventually, the container exploded.
Mixing incompatible chemicals can cause serious injuries and damage to property. Learn how safe hazardous chemical storage helps avoid such consequences.
Chemical Storage Guidelines: Understand the Chemical SDS
A Safety Data Sheet (SDS) comes with every chemical. All SDS follow a standard format governed by OSHA through their Hazard Communication Standard (HCS). Each SDS has 16 sections, detailed below. [Note: Sections 12 – 15 must be included on a SDS to be consistent with the United Nations’ Globally Harmonized System of Classification and Labeling of Chemicals (GHS). But, OSHA only mandates the headers. All content in these sections fall under other agencies’ jurisdictions.]
|Section||Name||Description||Status|
|1||Identification||Product or chemical name, including contact details for manufacturer||Mandatory|
|2||Hazard(s) Identification||Classes of hazards and appropriate warnings – includes labelling pictograms||Mandatory|
|3||Composition||Ingredients of compounds and mixtures and chemical identity||Mandatory|
|4||First-Aid Measures||Initial first-aid care instructions after exposure||Mandatory|
|5||Fire-Fighting Measures||Methods to extinguish a fire||Mandatory|
|6||Accidental Release Measures||Response measures for spills, leaks and releases||Mandatory|
|7||Handling and Storage||Guidance for handling and storage, including container compatibility issues||Mandatory|
|8||Exposure Controls / PPE||Measures to limit exposure and appropriate PPE for the specific chemical product||Mandatory|
|9||Physical and Chemical Properties||List of relevant properties, including appearance, odor, flammability, etc.||Mandatory|
|10||Stability and Reactivity||Descriptions of reactivity, chemical stability and indications of possible hazardous reactions||Mandatory|
|11||Toxicological Information||Description of toxicological and health effects of exposure||Mandatory|
|12||Ecological Information||Description of impact on environment in the case of release||Non-mandatory|
|13||Disposal Considerations||Advises on methods of disposal, recycling and reclamation of chemicals and containers||Non-mandatory|
|14||Transport Information||Classification information for transporting by road, air, rail or sea||Non-mandatory|
|15||Regulatory Information||Description of any relevant regulations specific to the product||Non-mandatory|
|16||Other Information||Revision history of SDS and other useful information||Mandatory|
Helpful Tip
Justrite supplies optional document storage boxes for safe SDS storage. Easily install boxes onto safety cabinet doors or side walls to protect important papers from moisture and dirt.
Chemical Storage Guidelines: Learn the Compatibility Issues Across Chemical Groups
It may seem obvious to store chemicals in alphabetic order for ease of use. But, this could result in storing incompatible chemicals right next to each other. Copper (II) sulfide reacts explosively with cadmium chlorate. A vigorous reaction occurs when hydrogen peroxide and iron (II) sulfide mix.
Chemical groups also react with other groups. Oxidizers are not directly flammable. But, they release oxygen, a key component of the fire triangle. It is risky storing them alongside flammable materials. Safety experts suggest a systematic approach to categorizing and storing chemicals.
Unfortunately, OSHA and the HCS requirements fail to provide one single method for chemical segregation and grouping. Many organizations have developed their own methods. Some of these are very complex. For instance, the U.S. Coast Guard uses 43 separate chemical groups to segregate chemicals for storage. But, simpler classifications exist that are comprehensive enough for most applications. Sciencelab, a leading chemical and laboratory equipment company, suggests these seven chemical groups:
- Flammables
- Oxidizers
- Acids
- Bases
- Highly reactives
- Extreme toxics / regulated materials
- Low hazard
The University of Iowa uses a similar breakdown with nine chemical groups:
- Flammable liquids
- Volatile poisons
- Oxidizing acids
- Organic and mineral acids
- Liquid bases
- Liquid oxidizers
- Non-volatile poisons
- Reactives
- Solids
No matter what organizational method is used for segregation, it is important to understand how these chemical groups react with one another and the associated risks.
Chemical Storage Guidelines: Tips for Safe Storage in a Safety Cabinet
- Don’t store chemicals in alphabetic order.
- Use shelf dividers and clear labeling for organization. Simple housekeeping and order goes a long way toward preventing human error, such as placing the wrong chemical in the wrong place.
- Only store hazardous chemicals in an approved flammable safety cabinet. Look for FM and UL approvals. These certify the cabinet meets OSHA and NFPA standards.
- Never store cardboard boxes in a flammable safety cabinet. Cardboard and paper may get saturated with flammable materials from spills and leaks. This creates a major fire risk.
- Always store flammable materials in a flammable storage cabinet, even if the chemical has other hazards. For example, aldehydes are both flammable and toxic. But, flammability trumps toxicity. Store aldehydes in a flammable storage cabinet. Secure it to prevent unauthorized access.
- Use clear signage on safety storage cabinets to identify its contents. Justrite safety cabinets feature exclusive Haz-Alert™ warning labels that inform all workers and first responders of its main chemical hazard – FLAMMABLE, ACID or HAZARDOUS. Justrite safety cabinets also come with a pack of 10 standard labels to identify specific contents.
- Never keep bottles of chemicals in a fume hood. Vapors from unsealed bottles could react with other chemicals in the fume hood, causing an explosion. Use Justrite under fume hood safety cabinets instead – both flammable and corrosive options available.
- Store non-flammable corrosive chemicals in polyethylene cabinets, such as a Justrite Polyethylene Corrosive and Acid Cabinet. These have no metal parts, since even corrosive fumes cause rust and reduce the life of your cabinet. Wooden laminate cabinets with a laminate finish are also suitable when storing corrosives.
Contact Justrite for Safe Hazardous Chemical Storage Solutions
Justrite specializes in flammable and hazardous chemical storage solutions. We offer a wide range of products for laboratory environments, including cabinets for corrosives and hazardous materials as well as safety containers for dispensing and HPLC disposal. All products meet and exceed codes and regulations. Browse all our safety cabinets or contact us to discuss your chemical segregation and storage needs. | https://www.justrite.com/news/cat/storage/post/chemical-storage-guidelines-for-safety-cabinets/ |
Stability: Stable. Incompatible with phosphorus, sulphur, zinc, ammonia, finely powdered metals, strong reducing agents, acids, organic materials.
Storage Condition: Keep away from heat, sparks, and flame. Do not store near combustible materials. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Description: Sodium chlorite (NaClO2) is a chemical compound used in the manufacturing of paper and as a disinfectant. | http://sparrowchemical.com/product_show.asp?id=1798 |
What is the law in terms of the storage of hazardous substances?
In many work places across the country, there are a vast amount of hazardous substances that, if not stored correctly can cause serious harm. Knowing how to correctly and safely store them is not only something that employers must follow to keep their staff safe but, it is also the law.
Hazardous substances can vary in type, hazard and severity as well as the conditions in which they are under by which they present a risk. Factors such as the amount of hazardous substance there is and how it is stored are points that must be considered when thinking about storing them safely.
There are a number of regulations in place to ensure the safe storage of hazardous substances such as the Health and Safety at work Act 1974 which includes a general duty requiring arrangements for the storage of substances to ensure the safety of those in the workplace.
Other regulations such as COSHH also require an employer to carry out a risk assessment including a consideration of how substances should be stored in order to keep everyone safe. Employers must provide adequate enough information, instruction and training for any employees who may use hazardous substances so that they are in accordance with a range of health and safety regulations.
As an employer, you must make sure that all substances are returned to their storage places when finished with, ensure all substances are stored safely in the first place and co-operate with their employees so that they comply with The Control of Substances Hazardous to Health Regulations 2002.
With substance types covering a wide range such as; flammable, oxidizing, lead, explosive, radioactive, oil, pesticides and substances hazardous to health, knowing the legislations for each is very important.
Factors to consider when safely storing hazardous substances include correctly labeling and packaging the components so that they can be correctly identified as well as taking into consideration; procedures for emergencies, the hazard presented by the substance, the amount of hazardous substance, the containers and how they will be stored.
Knowing how best to store your hazardous substances can really make all the difference when it comes to people safety. Storing dangerous materials correctly reduces the chance of accidents making working environments safer all around.
Examples as to how best to store hazardous substances are as follows. Gases should be stored in a safe, dry place on a flat surface and away from vehicles. Incompatibles such as acids should never be stored together and should be kept away from other substances. Highly flammable liquids and gases are best stored in closed tanks or cylinders and have security measures so that only authorised people can have access. Oil can be stored in tanks both above or under ground with one container not holding more than 200 litres in order to avoid water pollution.
Training for in the event of an emergency is always best practice when working with hazardous substances. Knowing that you have followed all correct health and safety procedures will ultimately keep everyone as safe as possible. | https://risk-assessment-products.co.uk/blog/what-is-the-law-in-terms-of-the-storage-of-hazardous-substances/ |
: R36/38- Irritating to eyes and skin.
immediately with plenty of water and seek S28- After contact with skin, wash immediately S37/39- Wear suitable gloves and eye/face S45- In case of accident or if you feel unwell, seek medical advice immediately (show the S61- Avoid release to the environment. Refer to special instructions/safety data sheet.
1-tetradecanamine, n,n-dimethyl-, n-oxide Other ingredients, determined not to be hazardous according to Safe Work Australia criteria, and not dangerous according to the ADG Code, make up the product concentration to 100%.
Other ingredients, determined not to be hazardous according to Safe Work Australia criteria, and not dangerous according to the ADG Code, make up the product concentration to 100%.
: No specific treatment. Treat symptomatically.
apparatus (SCBA) with a full face-piece operated in positive pressure mode.
: Immediately contact emergency personnel.
contact with soil, waterways, drains and sewers.
spill area. Approach the release from upwind.
section 1 for emergency contact information Note: see section 8 for personal protective equipment and section 13 for waste disposal.
equipment (see Section 8). Eating, drinking and smoking should be prohibited in areas where this material is handled, stored and processed.
Workers should wash hands and face before contaminated clothing and protective equipment before entering eating areas. Do not ingest.
Avoid breathing vapour or mist. Keep in the original container or an approved alternative made from a compatible material, kept tightly closed when not in use. Keep away from acids.
Empty containers retain product residue and can be hazardous. Do not reuse container.
Safe Work Australia (Australia, 8/2005).
: Use chemical-resistant, impervious gloves.
as necessary to prevent skin contact.
eating, smoking and using the lavatory and at the end of the working period.
Appropriate techniques should be used to remove potentially contaminated clothing.
Wash contaminated clothing before reusing. Ensure that eyewash stations and safety showers are close to the workstation location.
Product does not support combustion.
: The product is stable.
: Highly reactive or incompatible with the following materials: acids.
Reactive or incompatible with the following materials: metals.
: Under normal conditions of storage and use, hazardous reactions will not occur.
: Exposure to decomposition products may cause Product is sold ready to use.
Xi - IrritantR36/38- Irritating to eyes and skin.
: All substances are listed on AICS or exempt.
CONTINUING ACCURACY OF THIS INFORMATION. | http://medicpdf.com/i/inhospitality.com.au1.html |
PERSONAL PROTECTION :
Must wear eye protection glasses, mask, gloves and boots while handling the material.
FIRE FIGHTING METHODS :
Use dry chemical, CO2, sand, dolomite, etc. Water or halogenated extinguishing medium are NOT to be used.
HAZARD INFORMATION :
Avoid dust clouds formation, which may present a dust explosion hazard in the presence of a source of ignition.
HANDLING :
Keep away from heat, sparks and open flame. Containers to be kept closed tightly. This product will oxidize slowly if exposed to air.
PACKAGING :
MS Drums with double poly-lined bags inside. Silica gels inside the drums and inside the bags (optional).
STORAGE :
Store in a cool, dry, well-ventilated place and away from fire, sparks and incompatible substances.
SHELF LIFE :
Zinc Dust is sensitive to moisture. Unsealing the container and leaving exposed to open air or storing in humid areas will reduce the shelf life. | https://www.uniquesc.in/zinc.php |
Calcium bromide powder can be used to formulate the drilling, completion and workover fluids.
Technical Index
Appearance
White powder
Assay, %min
96.0
pH (5% solution)
6.5-9.5
Cl-, %max
0.5
SO42-, %max
0.05
Density of 52% water solution @20℃, g/cm3
1.70min
Water insoluble, %max
0.3
Moisture, %max
3.0
Heavy metal (Pb), ppm max
50
Recommended Handling
All personnel handling this material must handle it as an industrial chemical, wearing protective equipment and observing the precautions as described in the Material Safety Data Sheet.
Packaging and Storage
Packed in 25kg PP woven bag or 1 MT bulk bags.
Store in dry, well-ventilated area. Keep container closed. Keep away from heat, sparks and flames. Store away from incompatibles. Follow safe warehousing practices regarding palletizing, banding, shrink-wrapping and /or stacking. | http://uzhuchem.com/product/calcium-bromide-powder.html |
Product Name:
Name:
Octane,1-chloro- (Related Reference)
EINECS:
203-915-5
Molecular Formula:
C8H17Cl
CAS Registry Number:
111-85-3
Synonyms:
1-Octyl chloride; Capryl chloride; NSC 5406; Octyl chloride; n-Octyl chloride; n-Octyl chlorid;
InChI:
InChI=1/C8H17Cl/c1-2-3-4-5-6-7-8-9/h2-8H2,1H3
HS Code:
29031980
Molecular Structure:
This structure is also available as a 2d Mol file
Chemical Properties
Appearance:
Clear liquid
Molecular Weight:
148.67
Density:
0.874
Boiling Point:
183℃
Melting Point:
-61℃
Flash Point:
68℃
Storage Temperature:
Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Refractive index:
1.4288-1.4308
Solubility:
Insoluble
Stability:
Stable under normal temperatures and pressures.
Usage:
Barchlor(R) 8S is used as chemical intermediate.
Safety Data of 111-85-3
Risk Codes:
R52/53
Safety Statements: | http://www.chuanghuachem.com/product/showproduct.php?lang=en&id=458 |
Hazardous Materials3 min read
Hazardous materials (hazmat) are any material that has properties that may result in risk or injury to health and/or destruction of life or facilities. Many hazardous materials (hazmat) do not have a taste or an odor. Some can be detected because they cause physical reactions such as watering eyes or nausea. Some Hazardous Materials exist beneath the surface of the ground and have an oil or foam-like appearance. The substance can be identified from placards, labels or markings on the transporters.
Hazardous Material can be:
Corrosive Hazmat Materials:
are strong enough to eat away at steel drums or human skin. Because they can eat through the containers they are carried in, they are of special concern during transportation. Example: car battery acids
Ignitable Hazmat Materials:
present a fire hazard because they are flammable at relatively low temperatures. This causes a risk of explosion and the spreading of toxic gas over an area, as well as fire and smoke. Examples: paint removers, the chemical Benzene
Reactive Materials:
can explode or release deadly fumes by mixing with water or reacting to heat or pressure. Examples: old weapons and ammunition, sodium metal, stannic chloride
Radioactive Materials:
include materials with both high and low radioactivity that can lead to dangerous side effects for thousands of years. Most of these materials are produced at nuclear power plants and by research facilities.
Toxic Hazardous Materials:
consist of poisonous chemicals. People and animals exposed to these types of materials can develop severe health problems. Examples: lead, arsenic, mercury
Infectious Materials:
These materials are also toxic wastes, but are in a separate category. They consist of materials infected with some type of germ, bacteria, or virus that could cause disease in humans or animals. These types of materials often come from hospitals. Examples: hypodermic needles, human and animal waste.
Classification of Hazardous Materials:
The DOT has broad authority to regulate hazardous materials that are in transport, including the discretion to determine which materials shall be classified as “hazardous”. These materials are placed in one of nine categories, based on their chemical and physical properties. Based on the classification of the material, the DOT is also responsible for determining the appropriate packaging materials for shipping or transport. Finally, also based on the material classification, strict guidelines are furnished for proper labeling/marking of packages of hazardous materials offered for transport, and for placarding of transport vehicles.
Class 1: Explosives
Division 1.1 – Articles and substances having a mass explosion hazard
Division 1.2 – Articles and substances having a projection hazard, but not a mass explosion hazard
Division 1.3 – Articles and substances having a fire hazard, a minor blast hazard, and/or a minor projection hazard, but not a mass explosion hazard
Division 1.4 – Articles and substances presenting no significant hazard (explosion limited to package)
Division 1.5 – Very insensitive substances having a mass explosion hazard
Division 1.6 – Extremely insensitive articles which do not have a mass explosion hazard
Class 2 Gas
Division 2.1 – Flammable Gas
Division 2.2 – Non-flammable, non-toxic gas (under pressure, inert, etc.)
Division 2.3 – Toxic Gas
Class 3 Flammable Liquids (100 Degrees F or less closed cup):
Flammable Gas
Combustible
Class 4 Other Flammable Substances:
Division 4.1 – Flammable Solid
Division 4.2 – Substances liable to spontaneous combustion
Division 4.3 – Substances which, in contact with water, emit flammable gases
Class 5 Oxidizing Substances and Organic Peroxides:
Division 5.1 – Oxidizers
Division 5.2 – Organic Peroxides
Class 6 Toxic (Poisonous) and Infectious Substances:
Division 6.1 – Toxic Substances
Division 6.2 – Infectious Substances
Class 7 Radioactive Material:
Division 7 – Radioactive Material
Class 8 Corrosives: | https://hsseworld.com/hazardous-materials/ |
- Stability:Stable, but may form peroxides in storage if in contact with air. Highly flammable. Incompatible with oxidizing agents.
- CAS DataBase Reference628-81-9(CAS DataBase Reference)
- EPA Substance Registry SystemButane, 1-ethoxy- (628-81-9)
- Language:EnglishProvider:SigmaAldrich
- Language:EnglishProvider:ACROS
- Language:EnglishProvider:ALFA
Butyl ethyl ether Usage And Synthesis
- Chemical Propertiescolourless liquid
- Chemical PropertiesEthyl butyl ether is a colorless liquid
- UsesExtraction solvent, inert reaction medium.
- Synthesis Reference(s)The Journal of Organic Chemistry, 39, p. 3050, 1974 DOI: 10.1021/jo00934a027
- General DescriptionA clear colorless liquid with an ethereal odor. Flash point 40°F. Less dense than water. Vapors heavier than air.
- Air & Water ReactionsHighly flammable. Slightly soluble in water. Oxidizes readily in air to form unstable peroxides that may explode spontaneously [Bretherick 1979. p.151-154, 164]. A mixture of liquid air and diethyl ether exploded spontaneously [MCA Case History 616. 1960].
- Reactivity ProfileEthers, such as Butyl ethyl ether, can act as bases. They form salts with strong acids and addition complexes with Lewis acids. The complex between diethyl ether and boron trifluoride is an example. Ethers may react violently with strong oxidizing agents. In other reactions, which typically involve the breaking of the carbon-oxygen bond, ethers are relatively inert.
- HazardFlammable, dangerous fire risk.
- Health HazardInhalation or contact with material may irritate or burn skin and eyes. Fire may produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control may cause pollution.
- Fire HazardHIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water.
- Safety ProfileModerately toxic by ingestion. A skin and eye irritant. A very dangerous fire hazard when exposed to heat or flame; can react vigorously with oxidizing materials. Keep away from heat and open flame. To fight fire, use alcohol foam, CO2, dry chemical. When heated to decomposition it emits acrid smoke and irritating fumes. See also ETHERS.
- Potential ExposureUsed as a solvent for extraction and in making other chemicals
- ShippingUN1179 Ethyl butyl ether, Hazard Class: 3; Labels: 3-Flammable liquid.
- Purification MethodsPurify by drying with CaSO4, by passage through a column of activated alumina (to remove peroxides), followed by prolonged refluxing with Na and then fractional distillation. [Beilstein 4 IV 1518.]
- IncompatibilitiesMay form explosive mixture with air. Heat or prolonged storage may cause the formation of unstable peroxides. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, and epoxides. Attacks some plastics, rubber and coatings. May accumulate static electrical charges, and may cause ignition of its vapors.
- Waste DisposalDissolve or mix the material with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. All federal, state, and local environmental regulations must be observed. Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal.
- DL-Ethyl 2-bromobutyrate Diethyl 1,1-cyclopropanedicarboxylate ETHYL 4-CHLOROBUTYRATE Ethyl Succinyl Chloride 3-(2-FLUORO-PHENYL)-3-OXO-PROPIONIC ACID ETHYL ESTER Ethyl 4,4,4-trifluoroacetoacetate Heptafluorobutyric anhydride 4-Bromobutyl acetate Ethyl 2-bromohexanoate Ethyl 2-bromovalerate Ethyl 4-chloroacetoacetate ETHYL HEPTAFLUOROBUTYRATE Ethyl 2-bromoheptanoate Ethyl 6-bromohexanoate ETHYL 1-METHYLCYCLOPROPANE-1-CARBOXYLATE Ethyl cyclopropanecarboxylate 7-CHLOROCARBONYL-HEPTANOIC ACID ETHYL ESTER Ethyl 5-bromovalerate
Butyl ethyl etherSupplierMore
- Company Name:J & K SCIENTIFIC LTD.
- Tel:400-666-7788 010-82848833-
- Email:jkinfo@jkchemical.com;market6@jkchemical.com
- Company Name:Alfa Aesar
- Tel:400-610-6006
- Email:saleschina@alfa-asia.com
- Company Name:TCI (Shanghai) Development Co., Ltd. | https://m.chemicalbook.com/ProductChemicalPropertiesCB3854031_EN.htm |
Ocean Power Delivery’s (OPD) Pelamis wave energy converter is to undergo feasibility trials off the Scottish coast this summer.
The work is to take place at the Marine Energy Test Centre in Orkney and is to be carried out by OPD under a memorandum of understanding with AMEC and ScottishPower. The aim is to test the commercial and technical viability of the first full-scale device, construction of which was completed earlier this year.
The Pelamis generator is made up of four 24m canisters, aligned perpendicular to incoming waves. Energy is generated from the relative motion at the joints between each of the canisters. The maximum capacity of each device is 750kW. | https://www.nsenergybusiness.com/news/newssummer-tests-scheduled-for-pelamis-machine/ |
By Ahmed A. Shabana
Dynamics of Multibody platforms introduces multibody dynamics, with an emphasis on versatile physique dynamics. Many universal mechanisms resembling cars, house constructions, robots, and micro machines have mechanical and structural structures that include interconnected inflexible and deformable elements. The dynamics of those large-scale, multibody platforms are hugely nonlinear, featuring advanced difficulties that during so much circumstances can purely be solved with computer-based strategies. The e-book starts with a assessment of the fundamental principles of kinematics and the dynamics of inflexible and deformable our bodies sooner than relocating directly to extra complicated issues and computing device implementation. This re-creation contains vital new advancements in relation to the matter of enormous deformations and numerical algorithms as utilized to versatile multibody platforms. The book's wealth of examples and useful purposes may be priceless to graduate scholars, researchers, and training engineers engaged on a wide selection of versatile multibody structures.
Read Online or Download Dynamics of multibody systems PDF
Similar dynamics books
IUTAM Symposium on Nonlinear Stochastic Dynamics and Control: Proceedings of the IUTAM Symposium held in Hangzhou, China, May 10-14, 2010
Non-linear stochastic platforms are on the middle of many engineering disciplines and growth in theoretical study had resulted in a greater realizing of non-linear phenomena. This e-book offers info on new primary effects and their functions that are commencing to seem around the whole spectrum of mechanics.
Not like different books in this topic, which are inclined to be aware of 2-D dynamics, this article makes a speciality of the appliance of Newton-Euler how to advanced, real-life 3D dynamics difficulties. it's therefore perfect for non-obligatory classes in intermediate dynamics.
This ebook includes the lectures given on the moment convention on Dynamics and Randomness held on the Centro de Modelamiento Matem? tico of the Universidad de Chile, from December 9-13, 2003. This assembly introduced jointly mathematicians, theoretical physicists, theoretical computing device scientists, and graduate scholars attracted to fields with regards to likelihood conception, ergodic thought, symbolic and topological dynamics.
Nonequilibrium Carrier Dynamics in Semiconductors: Proceedings of the 14th International Conference, July 25–29, 2005, Chicago, USA
Foreign specialists assemble each years at this demonstrated convention to debate contemporary advancements in concept and test in non-equilibrium shipping phenomena. those advancements were the driver in the back of the brilliant advances in semiconductor physics and units during the last few a long time.
- Gas Dynamics of Thin Bodies
- Future Cities: Dynamics and Sustainability
- Nonlinear dynamics : from lasers to butterflies : selected lectures from the 15th Canberra International Physics Summer School, Australian National University, 21 January-1 February 2002
- Dynamics AX: A Guide to Microsoft Axapta
- Space Robotics: Dynamics and Control
Additional resources for Dynamics of multibody systems
Sample text
These kinematic constraints can be formulated by using a set of algebraic equations that imply that the relative translation between the two bodies along two axes perpendicular to the joint axis as well as the relative rotations between the two bodies must be zero. Similarly, the revolute joint, shown in Fig. 16(b), allows only relative rotation between the two bodies about an axis called the revolute joint axis. One requires five constraint equations: three equations that constrain the relative translation between the two bodies, and two equations that constrain the relative rotation between the two bodies to be only about the joint axis of rotation.
1) r, as shown in Fig. 2) where the vector b1 is drawn perpendicular to the plane OC Q¯ and thus has a direction (v × r¯ ), where v is a unit vector along the axis of rotation OC. The magnitude of the vector b1 is given by |b1 | = a sin θ From Fig. 3) The vector b2 in Eq. 2 has a magnitude given by θ 2 The vector b2 is perpendicular to v and also perpendicular to DQ, whose direction is the same as the unit vector (v × r¯ )/a. Therefore, b2 is the vector |b2 | = a − a cos θ = (1 − cos θ) a = 2a sin2 θ v × (v × r¯ ) θ · = 2[v × (v × r¯ )]sin2 2 a 2 Using Eqs.
However, as previously pointed out, the floating frame of reference formulation was mainly used in solving large reference displacement and small deformation problems. In Chapter 7 of this book, a conceptually different formulation called the absolute nodal coordinate formulation is presented. This formulation can be used efficiently for large deformation problems in flexible multibody system applications. 8 OBJECTIVES AND SCOPE OF THIS BOOK This book is designed to introduce the elements that are basic for formulating the dynamic equations of motion of rigid and deformable bodies. | http://realitat.com/index.php/pdf/dynamics-of-multibody-systems |
The Egyptian Tortoise Testudo kleinmanni (Critically Endangered) is, unlike its slow movement, declining fast. One of the world’s smallest tortoises, it occurs in the Mediterranean region and faces intense pressures. But regional cooperation gives it a gleam of hope.
The natural habitat of the Egyptian Tortoise is the desert and shrublands close to the coastal zone bordering Mediterranean Sea, where it has been recorded in Libya and Egypt. Nowadays, only small patches of territory are founded in Al Jabal Al Akhdar, the eastern coastal region of Libya, where it disappeared from much of its former range (and possibly a tiny population clings on in Zaranik Protected Area, Egypt).
Habitat loss, mainly due to agriculture, development, and other human activities, including the illegal trade, are the main reasons behind tortoise’s declines. Coastal development in the northern coastal region of Egypt degraded its natural habitat. Moreover, the political situation in Libya after the Libyan revolution in 2011 burdened the local communities and deteriorated the economic situation, while lack of stability and security on the Egyptian-Libyan border encouraged smugglers to enter the Libyan desert and collect tortoises to be sold in lucrative Egyptian pet markets. In Egyptian culture, this slow and peaceful creature is a symbol of luck and emblem of longevity and stability in life, making Egypt a good market for the tortoise. The market demand in Egypt caused their population to be harvested to local extinction, causing larger demand from its range in Libya.
Mature tortoises are more subjected to smuggling as a larger sized tortoise is more expensive, and unhealthy conditions during smuggling may causes serious infectious diseases among tortoises. Moreover, border guards were not aware that releasing the smuggled tortoises in the desert may cause the death of this peaceful creature.
Image credit: Egyptian Tortoise in Libya – © Al Hayat Organization
To overcome this transboundary issue and ensure the continuation of this species, conservation efforts must be taken at a regional level. Therefore, BirdLife (through its role as Regional Implementation Team for the Mediterranean Hotspot of the Critical Ecosystem Partnership Fund (CEPF)) provided small grants to two civil society organizations to assess Egyptian Tortoise populations in Eastern Libya and Western Egypt: Al Hayat Organization to Protect Wildlife and Marine Organisms (Libya) and Hemaya Company for Environmental Consultancies and Services (Egypt). Through these projects, remaining populations will be conserved in their original habitats in Libya and suitable habitats will be identified in Egypt for future reintroduction. This involves working together to identify in detail the main threats affecting the survival of the Egyptian Tortoise and assessing their ecological requirements.
“For more than 30 years, funding agencies did not provide any support to conserve this species”, says Basem Rabia, Egyptian Tortoise researcher. “Now, CEPF support and this fruitful cooperation gives me a hope that my dream will come true one day: of restoring the natural habitats of this wonderful small creature along the Mediterranean coast.”
“For more than 30 years, funding agencies did not provide any support to conserve this species. Now, CEPF support and this fruitful cooperation gives me a hope that my dream will come true one day: of restoring the natural habitats of this wonderful small creature along the Mediterranean coast.”
Basem Rabia, Egyptian Tortoise researcher
In Egypt, the team recorded some tortoises in tiny patches of territory within and around the Zaranik Protected Area. Studying the ecological requirements of this species is a must to ensure its survival, and so this remnant population gave researchers a good opportunity to study this species and its habitat. Meanwhile, in Libya, not many studies have been conducted on this species although it considered its natural habitat.
Hemaya Company conducted a survey in the northwestern Egypt to reassess the vegetation cover along the coast and identify the best sites for reintroduction. Seventy sites were assessed using on-ground site assessment and spatial modelling aimed at addressing the conservation needs for this species. Researchers thought that coastal development along the northern coast of Egypt caused an irreversible damage in the natural habitat, but healthy habitats, from 5 to 20 km south to the coast, showed they were wrong.
Al Hayat Organization, in turn, conducted a similar survey benefiting from the Egyptian experience at the Zaranik Protected Area to assess the vegetation cover and density that is necessary to the survival of the Egyptian Tortoise.
These surveys, along with various awareness raising sessions about the importance of Egyptian Tortoise to the ecosystem balance and the disadvantages of smuggling this species, showed local communities’ willingness to conserve this species. Moreover, a local community in Libya reported a tortoise smuggling attempt in 2021. As a result, the two grantees worked together with the Libyan Ministry of Environment to save 250 smuggled tortoises. After receiving these tortoises, Al Hayat Organization’s team sorted, classified, and marked them to help in tracing the turtles in the future. Later they released the tortoises in their natural habitat in Libya.
Through this cooperation, the two organizations will work in partnership to develop a joint action plan for future conservation of the Egyptian tortoise.
“Raising awareness about Egyptian tortoise and its smuggling is a critical issue in ensuring the sustainability of this species. This cooperation is a building block for future projects aiming at conserving Egyptian tortoises at larger scale.”
Murad Buijlayyil, Chairman of the Scientific Committee at Al Hayat Organization
*The Critical Ecosystem Partnership Fund (CEPF) is a joint initiative of l’Agence Française de Développement, Conservation International, the European Union, the Global Environment Facility, the Government of Japan, and the World Bank. Additional funding has been provided by the MAVA Foundation. A fundamental goal is to ensure civil society is engaged in biodiversity conservation.
CEPF is more than just a funding providerA dedicated Regional Implementation Team (RIT) (expert officers on the ground) guide funding to the most important areas and to even the smallest of organisations; building civil society capacities, improving conservation outcomes, strengthening networks and sharing best practices. In the Mediterranean Basin Biodiversity Hotspot, the RIT is entrusted to BirdLife International and its Partners: LPO (BirdLife France), DOPPS (BirdLife Slovenia) and BPSSS (BirdLife Serbia).Find out more at www.birdlife.org/cepf-med
Related news
Fishes, snails, plants, subterranean salamanders and more, the Mediterranean Basin Hotspot is globally important for its freshwater biodiversity. This vital habitat is of course vital for human life too, and as the demand for water increases, so can pressures on biodiversity.
We hear from four female conservation pioneers who have broken through gender stereotypes to pursue the careers they love – and benefit the whole community.
Bycatch from fishing is killing Europe’s seabirds in huge and unsustainable numbers. Jeremy Herry exposes the scale of the problem and sheds lights on the simple solutions that could turn things around – with enough political will.
Stay up to date
Our monthly newsletter curates the most fascinating articles across BLI’s work to save birds everywhere. | https://www.birdlife.org/news/2021/06/08/libyan-and-egyptian-conservationists-work-across-border-to-save-critically-endangered-tortoise/ |
The boreal felt lichen (Erioderma pedicellatum) is considered critically endangered by the International Union for Conservation of Nature. Although the species has suffered major reductions in parts of its range, new findings in Alaska, Russia, and Newfoundland have significantly increased the estimated population and geographic range. The Newfoundland population is particularly interesting because some areas are showing rapid declines while other areas are either showing increase or relatively stable populations. We sampled populations of the boreal felt lichen in plots systematically placed along transects in a ribbed moraine landscape. Our intent was to capture as much of the ecological variation in order to better understand the population structure of this species in different habitats and to develop a predictive habitat model to assist with strategic land-use planning. We found 668 thalli on 375 trees. The vast majority of the boreal felt lichen thalli were on balsam fir trees between 1 and 2 m in height. The number of thalli per tree varied between 1 and 19. We observed several factors that are likely to have an influence on the dynamics of the boreal felt lichen populations including competition from chlorolichens, mortality of the phorophyte, and physical damage from falling trees. This information combined with a detailed ecosystem mapping project will help in the development of a predictive habitat model. We will discuss further the dynamics of the boreal felt lichen populations in the context of a changing forest ecosystem and the management implications for an endangered species in a managed landscape. | https://cfs.nrcan.gc.ca/publications?id=36316 |
Kelly Fitzgerald-HollandSenior Wildlife Biologist & Regulatory Specialist
Kelly is a Certified Wildlife Biologist, senior wildlife biologist and environmental compliance expert. She has over 20 years of experience in ecological research, program management, environmental regulation and compliance, and terrestrial ecosystem monitoring in the western U.S. She has served as senior wildlife biologist or task lead manager for a large number of projects that require endangered species permitting and biological analysis for CEQA/NEPA compliance. Kelly specializes in evaluating impacts on threatened and endangered wildlife species and their habitats, coordinating with resource agency staff to ensure compliance with the Federal and State Endangered Species Acts, including completing Section 7 consultation, and assisting clients with developing compensatory habitat mitigation and planning for long-term management of the habitat. Prior to her position at GEI, she spent 4 years conducting ESA consultations as a USFWS biologist, reviewing projects to assess impacts on listed species, providing technical assistance to minimize impacts on listed species, and preparing biological opinions for projects that impacted federally listed species and designated critical habitat. While at USFWS, Kelly garnered extensive knowledge of the federally threatened giant garter snake, assisting with conservation and recovery planning for this species through research consolidation, technical oversight and coordination with species experts. In addition to having worked for USFWS, as well as the National Park Service, U.S. Forest Service and the Peace Corps, Kelly has worked extensively with state and federal agencies to assist clients with compliance with CESA/ESA, Migratory Bird Treaty Act, and CEQA/NEPA.
Kelly has a M.S. in Environmental Science from Washington State University – Pullman and a B.A. in Environmental Studies from the University of California – Santa Cruz. | https://www.geiconsultants.com/?team=kelly-fitzgerald-holland |
This is a case study that's a part of the 2021 REIT Industry ESG Report, an annual report detailing the REIT industry's environmental, social, and governance (ESG) performance details in the publicly traded U.S. REIT industry. Featured case studies showcase REIT leadership and ESG innovation from a variety of sectors and serve as a practical tool for stakeholders to assess the scale and impact of the REIT industry's ESG commitments and initiatives.
Equity LifeStyle Properties' (ELS) portfolio spans over 39,000 acres— encompassing 32 threatened and endangered species in critical habitats, about 8,000 forested acres, and over 2,200 acres of wetlands.
As such, environmental topics including Water Consumption and Conservation, Energy Consumption and Efficiency, Climate Change Mitigation and Adaptation, and Biodiversity are of material importance to ELS—and have therefore inspired a number of environmental projects.
In particular, the De Anza Santa Cruz Restoration Project at a 198-site manufactured home community on the shores of the Pacific Ocean serves as an illustrative example of ELS' environmental commitments.
"My favorite part of the De Anza Santa Cruz Restoration Project is that now we see so many more birds from the bridge. I was involved in the annual Audubon Christmas bird count for my area of De Anza Santa Cruz, particularly around the ponds. We spotted 31 species in two hours, which is truly phenomenal for a place this small."
Jenny Anderson, three-year resident of the De Anza Santa Cruz community
ELS completed a large-scale, multi-phase ecological and environmental conservation and restoration project at the property, working closely with the California Coastal Commission, California Department of Fish and Wildlife, and California Department of Housing and Community Development.
The wide-ranging scope of the project included:
- Improvements to the community's stormwater infrastructure
- Restoration of the plant habitat surrounding the community ponds
- Removal of invasive species
- Creation of new habitats designed to attract native wildlife and endangered species
- Development of dedicated spaces—such as vista overlooks, gardens, and pathways—that provide peaceful outdoor areas for residents along wildlife habitats
- Incorporation of locally sourced native plants to help reduce the project's carbon footprint
The project has been particularly appreciated by residents of the community who enjoy seeing the growth in biodiversity.
By pursuing additional environmental stewardship projects in the future, ELS is well positioned to continue maintaining biodiversity across its portfolio, creating assets that are connected to their local and natural environments, and providing places where residents and guests can enjoy and unite with nature.
Equity LifeStyle Properties is a leading owner and operator of manufactured home communities, RV resorts and campgrounds, and marinas in North America. | https://www.reit.com/news/blog/nareit-developments/equity-lifestyle-properties-maintaining-biodiversity-and-benefiting |
The Market Strategy Senior Consultant role within New Business Innovation's Hybrid Business Design team is responsible for contributing to the execution of market analyses that inform hybrid business cases and investment decisions. This position will work with teammates to deliver services, including testing key hypotheses in-market, evaluating competitive landscapes, and ultimately making actionable recommendations that will shape net-new or early-stage hybrid business ideas.
The position requires capabilities at both the strategy and execution levels. The candidate will be expected to maintain expertise in current market analyses trends and techniques, including: market fit analysis, market sizing, competitive scanning, pricing validation, partnering analyses, voice of the customer insight synthesis, and interview techniques (both internal and external). Additionally, the candidate should be able to think beyond the data collected to distill and present meaningful insights that can be translated to action-oriented recommendations for senior executive leadership within Deloitte.
The candidate will also have the opportunity to work with other team members across Pricing and Ecosystem partnerships to expand their skills and better understand implications to business decisions Deloitte is making.
Specific essential functions of this role include:
Execute day-to-day development of multiple market assessments at once
Collaborate with technical and non-technical stakeholders at varying levels to deeply understand the market size and demand, competitive landscape, and partnering opportunities for high-growth, hybrid business cases and investment decisions
Support leaders as they develop key hypotheses to iteratively test through secondary and primary research
Identify and understand competitive offerings, including key capabilities
Perform market sizing estimation and detailed competitive analyses to validate or evolve Network stakeholders' point of view, including lead and/ or support Total Addressable Market (TAM) analysis
Analyze key industry trends to develop a strategic point of view on where a market is in the context of specific offerings
Maintain an informed point of view on new and emerging business models
Maintain an informed point of view on emerging technologies and related services
Prepare and conduct external interviews with both target clients and key competitors (both at the executive level) to test key assumptions on clients' business challenges, perceived value drivers, and critical buying factors as well as competitors' differentiation
Synthesize large amounts of secondary research and interview data to develop key insights that can inform investment decisions for technical assets
Prepare and lead executive level analysis output and presentations
Act as POC for respective area on a project-by-project basis
The Team: Hybrid Business Design
The Hybrid Business Design team within Deloitte Consulting is primarily responsible for enabling new hybrid businesses by architecting and orchestrating offerings that pioneer differentiated business models for Deloitte Consulting.
We are looking for individuals who can help test, identify, and recommend the highest potential business ideas by collaborating with the Network, ecosystem alliances, and other enabling groups to shape the partnership component of these new concepts.
The group is part of Deloitte Consulting's New Business Innovation team, which focuses on scaling Consulting's asset-based agenda to deliver tangible results in the near-term and a sustainable competitive advantage for the firm in years to come.
Qualifications
Required:
Bachelor's Degree in Business, Finance, Marketing, Economics or a related field
4+ years of experience in business strategy and business case development, including market analysis, interviewing skills, and data distillation to insights preferred
2+ year experience with asset-enabled business, including development software business cases and / or delivering technology-based projects
Preferred:
Ability to support and conduct internal and external interviews with executive-level experts, including synthesizing interview notes to develop key insights
Experience in developing business cases for technology-based solutions, including analyses like: market sizing, competitor landscape, voice of the customer, pricing, cross-offering portfolio opportunities, and technical build recommendations.
Experience in applying standard methods for establishing and testing addressable market size estimates
Proven ability to successfully partner with internal senior stakeholders from various parts of the organization, with varying degrees of technical aptitude, and drive successful outcomes cross-functionally
Ability to develop high-quality deliverables with limited input from executive sponsors
Strong verbal and written communication skills
Ability to juggle many competing priorities/ deadlines
Understanding of Deloitte Consulting's organization, including matrixed nature and Offering Portfolios
*Must be legally authorized to work in the United States without the need for employer sponsorship, now or at any time in the future.
The wage range for this role takes into account the wide range of factors that are considered in making compensation decisions including but not limited to skill sets; experience and training; licensure and certifications; and other business and organizational needs. The disclosed range estimate has not been adjusted for the applicable geographic differential associated with the location at which the position may be filled. At Deloitte, it is not typical for an individual to be hired at or near the top of the range for their role and compensation decisions are dependent on the facts and circumstances of each case. A reasonable estimate of the current range is $85,000 to $155,000.
You may also be eligible to participate in a discretionary annual incentive program, subject to the rules governing the program, whereby an award, if any, depends on various factors, including, without limitation, individual and organizational performance.
#NBI22
#Product22
#LI-XL1
#LI-Remote
All qualified applicants will receive consideration for employment without regard to race, color, religion, sex, sexual orientation, gender identity, national origin, age, disability or protected veteran status, or any other legally protected basis, in accordance with applicable law.
All qualified applicants will receive consideration for employment without regard to race, color, religion, sex, sexual orientation, gender identity, national origin, age, disability or protected veteran status, or any other legally protected basis, in accordance with applicable law. | https://jobsus.deloitte.com/tampa-fl/sr-market-strategy-consultant-nbi_us/439A5B37C6734E5FA47BE0FCD764F19D/job/?vs=28 |
Source The photograph that has become known as "Migrant Mother" is one of a series of photographs that Dorothea Lange made in February or March of in Nipomo, California. InLange gave this account of the experience: I saw and approached the hungry and desperate mother, as if drawn by a magnet.
Onset of depression more complex than a brain chemical imbalance Updated: April 11, Published: June, It's often said that depression results from a chemical imbalance, but that figure of speech doesn't capture how complex the disease is.
Research suggests that depression doesn't spring from simply having too much or too little of certain brain chemicals. Rather, there are many possible causes of depression, including faulty mood regulation by the brain, genetic vulnerability, stressful life events, medications, and medical problems.
It's believed that several of these forces interact to bring on depression. To be sure, chemicals are involved in this process, but it is not a simple matter of one chemical being too low and another too high.
Rather, many chemicals are involved, working both inside and outside nerve cells. There are millions, even billions, of chemical reactions that make up the dynamic system that is responsible for your mood, perceptions, and how you experience life.
With this level of complexity, you can see how two people might have similar symptoms of depression, but the problem on the inside, and therefore what treatments will work best, may be entirely different. Researchers have learned much about the biology of depression. They've identified genes that make individuals more vulnerable to low moods and influence how an individual responds to drug therapy.
One day, these discoveries should lead to better, more individualized treatment see "From the lab to your medicine cabinet"but that is likely to be years away. And while researchers know more now than ever before about how the brain regulates mood, their understanding of the biology of depression is far from complete.
What follows is an overview of the current understanding of the major factors believed to play a role in depression. The brain's impact on depression Popular lore has it that emotions reside in the heart.
Science, though, tracks the seat of your emotions to the brain. Certain areas of the brain help regulate mood. Researchers believe that — more important than levels of specific brain chemicals — nerve cell connections, nerve cell growth, and the functioning of nerve circuits have a major impact on depression.
Still, their understanding of the neurological underpinnings of mood is incomplete. Regions that affect mood Increasingly sophisticated forms of brain imaging — such as positron emission tomography PETsingle-photon emission computed tomography SPECTand functional magnetic resonance imaging fMRI — permit a much closer look at the working brain than was possible in the past.
An fMRI scan, for example, can track changes that take place when a region of the brain responds during various tasks. Use of this technology has led to a better understanding of which brain regions regulate mood and how other functions, such as memory, may be affected by depression.
Areas that play a significant role in depression are the amygdala, the thalamus, and the hippocampus see Figure 1. Research shows that the hippocampus is smaller in some depressed people. For example, in one fMRI study published in The Journal of Neuroscience, investigators studied 24 women who had a history of depression.
The more bouts of depression a woman had, the smaller the hippocampus. Stress, which plays a role in depression, may be a key factor here, since experts believe stress can suppress the production of new neurons nerve cells in the hippocampus.
Researchers are exploring possible links between sluggish production of new neurons in the hippocampus and low moods.
An interesting fact about antidepressants supports this theory. These medications immediately boost the concentration of chemical messengers in the brain neurotransmitters. Yet people typically don't begin to feel better for several weeks or longer. Experts have long wondered why, if depression were primarily the result of low levels of neurotransmitters, people don't feel better as soon as levels of neurotransmitters increase.
The answer may be that mood only improves as nerves grow and form new connections, a process that takes weeks. In fact, animal studies have shown that antidepressants do spur the growth and enhanced branching of nerve cells in the hippocampus. So, the theory holds, the real value of these medications may be in generating new neurons a process called neurogenesisstrengthening nerve cell connections, and improving the exchange of information between nerve circuits.
If that's the case, medications could be developed that specifically promote neurogenesis, with the hope that patients would see quicker results than with current treatments. Areas of the brain affected by depression Amygdala: The amygdala is part of the limbic system, a group of structures deep in the brain that's associated with emotions such as anger, pleasure, sorrow, fear, and sexual arousal.
The amygdala is activated when a person recalls emotionally charged memories, such as a frightening situation. Activity in the amygdala is higher when a person is sad or clinically depressed. This increased activity continues even after recovery from depression. The thalamus receives most sensory information and relays it to the appropriate part of the cerebral cortex, which directs high-level functions such as speech, behavioral reactions, movement, thinking, and learning.How your immune system causes depression.
Chapter 1 Introduction. In I wrote a speculative biomedical paper suggesting that the immune system is the key to understanding a broad range of mental illnesses, including depression, schizophrenia, hyperactivity, anxiety and manic-depressive disorder.
1 The basic idea was this: the . The causes of stress in modern life emerge from the many obligations we have to handle everyday to the modern way of negative thinking. We have all experienced stress periods in our life.
This is something normal. Lost Connections: Uncovering the Real Causes of Depression [Johann Hari] on yunusemremert.com *FREE* shipping on qualifying offers.
From the New York Times bestselling author of Chasing the Scream, a radically new way of thinking about depression and anxiety What really causes depression and anxiety - and how can we really solve them? Award-winning journalist Johann Hari suffered from depression.
Psychotherapy. There is a wide number of effective therapeutic approaches utilized for the treatment of depression today. These range from cognitive behavioral therapy, to behavioral therapy (e.g.
The causes of the Great Depression in the early 20th century have been extensively discussed by economists and remain a matter of active debate. They are part of the larger debate about economic yunusemremert.com specific economic events that took place during the Great Depression are well established.
There was an initial stock market crash that .
Poems About Depression and Suicide offers heartfelt poetry on a wide range of topics ranging from melancholy to suicide. | https://mowuhab.yunusemremert.com/the-different-causes-of-depression-in-modern-world-12819bf.html |
The Science Of First Impressions: Emotions Influence Social Parts Of The Brain To Work Differently
Do you remember the first time you met your best friend, or saw the love of your life? A new study, published in the journal ELife, pulls the veil back on the brain to demonstrate how we process those initial encounters into memories. Researchers from the University of Haifa in Israel examined the emotional impact of the first impression, revealing it to be a key player in creating memories.
“It turns out that different emotions cause the brain to work differently and on distinct frequencies,” said the study’s lead author Dr. Shlomo Wagner, a neuroscientist at U-Haifa, in a press release. “We found a connection between the feeling of excitement, rhythmical activity in specific brain areas, and the cognitive process of memory formation. In essence, this finding explains why people tend to remember in particular their first encounter with a future friend or partner.”
In the first stage of the experiment, Wagner and his research team studied rats and traced the areas of the brain that lit up as they socialized with other rats. While rats' brains lit up with excitement when they encountered other rats for the first, researchers were surprised to see the memory-forming part of their brain lit up simultaneously. When the rats met for a second time, the brain activity in the two areas declined.
Comparing the two different encounters, researchers found the brain initially worked “intensely” under a unique communication system in order to coordinate the two areas of the brain. But as the animals got to know each other better, the brain’s emotional connectivity weakened. It turns out the carefully calibrated synchronized system of electrical signals is dictated by those first emotions.
The rat stranger elicited high levels of excitement in the rat’s brain, ultimately facilitating the formation of social memory. But to test their emotions, researchers frightened the rats with other rats, and saw that the brain worked under a different communication system.
When a negative emotion like fear is the initial reaction instead of excitement, the brain turns on different electrical signals synchronized with memory formation. It scientifically validates the idea you never get a second chance to make a first impression. "It seems that when the emotion is social and positive, the brain tells the different areas to work according to one communication protocol," Wagner said.
Earlier this year, a team of researchers at New York University’s psychology department discovered the brain’s ability to retrieve memories was improved when associated with emotions. They also found different regions in the brain were influenced by different emotions and that overtime, memories were strengthened. The emotional impact of memories and new experiences may explain why certain emotions progressively develop in the brain.
“We will need to conduct additional studies in the future in order to understand the precise ramifications of each emotion on memory,” Wagner said. “But in broad terms the implication is clear. Different emotions cause the brain to work differently, including in terms of cognitive processes such as learning and memory.”
Source: Wagner S and Tendler A. Different types of theta rhythmicity are induced by social and fearful stimuli in a network associated with social memory. eLife. 2015. | https://www.medicaldaily.com/science-first-impressions-emotions-influence-social-parts-brain-work-differently-346846 |
We are all dealing with this pandemic in our own way as we grapple through unchartered territory. Now, more than ever, in the midst of extreme uncertainty and anxiety, we need to find ways to put in practice what we have always known – that the answer to our quest for happiness lies within ourselves.
At a time when most of our coping mechanisms (such as meeting friends over a meal, shopping, going to the gym or talking a long walk) are not available, it becomes doubly difficult to not be angry, irritable and frustrated, leave alone feel contentment or happiness. But more than in normal times, it is imperative for the well-being of our loved ones and ourselves that we find a way to manage our feelings and consciously make an effort to increase our happy and feel-good hormones. Our feelings, whether they are of well-being, joy and peace or whether they are of grief, depression and anger – are all regulated by hormones and neurotransmitters. Neurotransmitters are brain chemicals that communicate information between the brain and body. They relay signals between the neurons or nerve cells. These important chemicals interact with receptors located throughout the brain and the body to regulate a wide variety of processes including emotions, fear, pleasure, joy, anger, mood and the perception of pain. Emotional intelligence lies in our ability to become aware of our inner state and understand how to make our emotions work for us, rather than allowing them to rule over our minds.
There are many chemicals that influence our feelings but they can be narrowed down to four main neurotransmitters which play an important role in feeling a sense of well-being or happiness:
Dopamine – drives our brains reward system and its production is increased when we seek out ways to find pleasure or when we achieve an objective or goal. | https://www.magzter.com/article/Lifestyle/GlobalSpa/Keep-up-the-HAPPY-HORMONES-in-this-unprecedented-time |
Can you see better when you exercise?
Participants rode stationary bikes while wearing a wireless heart rate monitor and an EEG cap.
Intrigued by recent findings that neuron firing rates in the regions of mouse and fly brains associated with visual processing increase during physical activity, UC Santa Barbara psychologists Barry Giesbrecht and Tom Bullock wanted to know if the same might be true for the human brain.
To find out, they designed an experiment using behavioral measures and neuroimaging techniques to explore the ways in which brief bouts of physical exercise impact human performance and underlying neural activity. The researchers found that low-intensity exercise boosted activation in the visual cortex, the part of the cerebral cortex that plays an important role in processing visual information. Their results appear in the Journal of Cognitive Neuroscience.
“We show that the increased activation — what we call arousal — changes how information is represented, and it’s much more selective,” said co-author Giesbrecht, a professor in UC Santa Barbara’s Department of Psychological and Brain Sciences. “That’s important to understand because how that information then gets used could potentially be different.
To investigate how exercise affects different aspects of cognitive function, the investigators enlisted 18 volunteers. Each of them wore a wireless heart rate monitor and an EEG (electroencephalogram) cap containing 64 scalp electrodes. While on a stationary bicycle, participants performed a simple orientation discrimination task using high-contrast stimuli composed of alternating black and white bars presented at one of nine spatial orientations. The tasks were performed while at rest and during bouts of both low- and high-intensity exercise.
The scientists then fed the recorded brain data into a computational model that allowed them to estimate the responses of the neurons in the visual cortex activated by the visual stimuli. They analyzed the responses while participants were at rest and then during low- and high-intensity exercise.
This approach allowed them to reconstruct what large populations of neurons in the visual cortex were doing in relation to each of the different stimulus orientations. The researchers were able to generate a “tuning curve,” which estimates how well the neurons are representing the different stimulus orientations.
Giesbrecht noted that they don’t know the mechanism by which this is occurring. “There are some hints that it may be driven by specific neurotransmitters that increase global cortical excitability and that can account for the change in the gain and the increase in the peak response of these tuning profiles,” he said.
From a broader perspective, this work underscores the importance of exercise. “In fact, the benefits of brief bouts of exercise might provide a better and more tractable way to influence information processing — versus, say, brain training games or meditation — and in a way that’s not tied to a particular task,” Giesbrecht concluded. | https://www.universityofcalifornia.edu/news/can-you-see-better-when-you-exercise |
Exciting advances in neuroscience have taken place during the past decade.
A number of them relate to a better understanding of neuropsychiatric diseases. Equally important progress is also being made in our knowledge of the neurobiology of positive states and traits including well-being, resilience, and wisdom.
Based on neuroimaging, genetic, and animal studies, specific brain regions and neurotransmitters have been identified as being critical to the development of these characteristics. In foreseeable future, it may become possible to use behavioral and biological techniques to enhance positive traits and states in people suffering from specific neuropsychiatric disorders. | https://www.cbr.iisc.ac.in/lectures-01.htm |
Multiple things can contribute to depression. These include stressful situations, a lack of support, and life events like losing a loved one or a job. Additionally, some individuals suffer from depression owing to specific medical conditions. Depression is more prevalent among individuals with chronic illnesses such as diabetes, cancer, Parkinson's disease, or other conditions. Additionally, alcohol and recreational substances can exacerbate sadness. Lastly, individuals who are easily overwhelmed may also suffer from depression. Changes in hormone levels produce mood swings. Due to these changes, which may occur during menstruation, pregnancy, or menopause, your mood may fluctuate. An underlying mental health disorder could also cause mood swings. If you have regular mood fluctuations, you should consult a physician. You might be able to manage these symptoms with the help of other therapy and mood stabilizers. Those affected by these challenges who wish to develop more robust coping mechanisms can benefit of psychotherapy.
It is recognized that stress, traumatic experiences, and hormonal changes can all produce mood swings. Mood swings can have a negative influence on relationships and workplace efficiency, and they might be an indicator of a mental health issue. This article will examine the causes and various therapies for mood swings. Many people regularly endure mood fluctuations. Although this is typical, mood fluctuations that interfere with daily life may signal a more severe condition. If your mood fluctuations are too frequent or interfere with your everyday activities, you should visit a physician. If you or someone you care about has sustained a brain injury, you should consult a mental health expert. Those who have suffered a traumatic brain injury (TBI) are significantly more prone than those who have not acquired depression. There are numerous solutions to this problem, which is fortunate.
The most frequent head injury is a concussion, which occurs when the brain is jolted and strikes the skull. Depending on the cause, the severity of trauma can range from moderate to severe. They may also be induced by a strike to a different body part. Other head injuries include intracranial hematomas, bleeding inside the skull, contusions, or bruises on the brain. Call 911 immediately if you have sustained a head injury. Even if there are no apparent symptoms, moving a person with an injury may exacerbate their condition. The Glasgow Coma Scale, a 15-point scale used to determine injury severity, is utilized by medical workers trained to transport injured patients with care. Although scientists are unaware of how chemical imbalances in brain produce depression, they are confident that certain antidepressants effectively treat the illness in some patients. However, even though these medications enhance serotonin levels in the brain, it is debatable whether they benefit everyone. In truth, numerous individuals benefit from drugs with distinct mechanisms of action.
Although there is no single cause of depression, several factors enhance the risk. For instance, a vitamin C shortage and persistent inflammation might contribute to depression. On the other hand, a diet rich in vitamins C and E can aid in the prevention of depressive symptoms due to their association with hormonal imbalances. Studies have connected depression to additional molecular brain pathways. Examples include the opioid, glutaminergic, and cholinergic systems. In addition, studies have found that the relative amounts of these neurotransmitters in various brain regions can influence depression. SAD is a prevalent condition that causes people to experience seasonal depression and irritability. All ages are impacted. However, adolescents and women are afflicted more severely than men. Although the exact cause is unknown, fewer daylight hours are believed to be responsible. In addition, some individuals inherit genes that raise their likelihood of having this illness. SAD is treatable with both psychotherapy and medicine.
Leaving the house helps alleviate SAD symptoms. You can increase the natural light entering your home by opening your drapes and trimming neighboring trees. To avoid feeling lonely and angry, make an effort to be physically and socially active; schedule fun activities.
Various treatments exist for SAD, including medication and light therapy. However, only brief alleviation is provided by these procedures. Most patients begin treatment within two weeks. However, it may take longer in some cases. Light therapy should ideally start as soon as a person rises in the morning. Cognitive behavioral therapy is an alternative treatment for SAD. It helps patients to monitor their emotions and exercise self-control, which may lessen the severity of their SAD symptoms during their most severe episodes. | https://6283768275a38.site123.me/blog/the-leading-four-causes-of-depression-are-as-follows |
1) It is recommended that a veterinarian or licensed veterinary technician supervise the patient admissions.
a) A properly trained nonprofessional can be quite capable of handling this process but we recommend that a licensed professional be available to assist this individual should questions or concerns arise.
2) The patient’s medical record should be reviewed for completeness.
3) A pre-surgical review of the patient’s history should be performed prior to admission. An admission checklist or questionnaire can be a valuable tool to insure a thorough assessment of the patient. An example questionnaire is included at the end of this reference.
a) Food should be withheld for 12 hours prior to admission in normal cats and dogs over age 4 months.
i) For cats and dogs under 4 months of age, hypoglycemia is a significant concern.
(1) Withholding food for only 4 hours prior to anesthesia helps to minimize this concern.
(2) Offering food within 2 or 3 hours of recovery is also recommended.
b) Any medications or supplements given in the prior 7 days should be recorded and reviewed with a doctor.
c) Any new health concerns should be recorded and reviewed with a doctor.
d) Any previous anesthetic “problems” should be reviewed with owner, recorded and reviewed with a doctor.
e) Pets with histories of excessive stress when kenneled, chewing at stitches or bandages, difficulty restricting activity, or difficulty maintaining pet in clean, dry area during recovery should be noted.
f) Water should not be withheld prior to admission.
4) Smaller growths should be marked for easy identification.
a) The owner should show the admitting staff member where the growths are, and the staff member should mark them with the owner present.
i) Hair can be clipped at the site or a marker used to identify the site.
5) An accurate Estimate and Surgical Release Form should be reviewed with, and signed by, the owner.
a) Please be vigilant for owners who may not understand the form or, in fact, may not be able to read the form.
i) A resistance to sign the form may be one indication of this.
|Return to top of page|
|Questions or problems regarding this web site should be directed to DRSTEIN@VASG.ORG .
|
Copyright © 2003 ASAH. All rights reserved.
Last modified: April 9, 2011 . | https://vasg.org/admissions.htm |
Knowledge Pile is a knowledge-related data leveraging Query of CC.
This dataset is a partial of Knowledge Pile(about 40GB disk size), full datasets have been released in [🤗 knowledge_pile_full], a total of 735GB disk size and 188B tokens (using Llama2 tokenizer).
Query of CC
Just like the figure below, we initially collected seed information in some specific domains, such as keywords, frequently asked questions, and textbooks, to serve as inputs for the Query Bootstrapping stage. Leveraging the great generalization capability of large language models, we can effortlessly expand the initial seed information and extend it to an amount of domain-relevant queries. Inspiration from Self-instruct and WizardLM, we encompassed two stages of expansion, namely Question Extension and Thought Generation, which respectively extend the queries in terms of breadth and depth, for retrieving the domain-related data with a broader scope and deeper thought. Subsequently, based on the queries, we retrieved relevant documents from public corpora, and after performing operations such as duplicate data removal and filtering, we formed the final training dataset.
Knowledge Pile Statistics
Based on Query of CC , we have formed a high-quality knowledge dataset Knowledge Pile. As shown in Figure below, comparing with other datasets in academic and mathematical reasoning domains, we have acquired a large-scale, high-quality knowledge dataset at a lower cost, without the need for manual intervention. Through automated query bootstrapping, we efficiently capture the information about the seed query. Knowledge Pile not only covers mathematical reasoning data but also encompasses rich knowledge-oriented corpora spanning various fields such as biology, physics, etc., enhancing its comprehensive research and application potential.

This table presents the top 10 web domains with the highest proportion of Knowledge Pile, primarily including academic websites, high-quality forums, and some knowledge domain sites. Table provides a breakdown of the data sources' timestamps in Knowledge Pile, with statistics conducted on an annual basis. It is evident that a significant portion of Knowledge Pile is sourced from recent years, with a decreasing proportion for earlier timestamps. This trend can be attributed to the exponential growth of internet data and the inherent timeliness introduced by the Knowledge Pile.
| Web Domain | Count |
|---|---|
| en.wikipedia.org | 398833 |
| www.semanticscholar.org | 141268 |
| slideplayer.com | 108177 |
| www.ncbi.nlm.nih.gov | 97009 |
| link.springer.com | 85357 |
| www.ipl.org | 84084 |
| pubmed.ncbi.nlm.nih.gov | 68934 |
| www.reference.com | 61658 |
| www.bartleby.com | 60097 |
| quizlet.com | 56752 |
cite
@article{fei2024query,
title={Query of CC: Unearthing Large Scale Domain-Specific Knowledge from Public Corpora},
author={Fei, Zhaoye and Shao, Yunfan and Li, Linyang and Zeng, Zhiyuan and Yan, Hang and Qiu, Xipeng and Lin, Dahua},
journal={arXiv preprint arXiv:2401.14624},
year={2024}
}
- Downloads last month
- 95